
|
 |

|

|
|

|
|
Thank Yous
(coming Soon!)
|
|
# 9 - Noble GasesLook at the last row on the right of the periodic table:
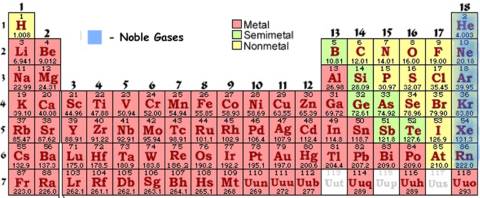
These elements are called the noble gasses. What�s so special about them to get its own lesson? These atoms already have eight in their outer shell. Remember the octet rule? The octet rule sates that atoms achieve "lower energy" by having 8 electrons in their outer shell.
Helium (the exception noted above), has only two electrons in it�s outer shell, but it�s following the duet rule. That means it is at "lower energy" by having 2 electrons. It�s all it can fit! |

