|
 |

|

|
|

|
|
Thank Yous
(coming Soon!)
|
|
#6 - Atom Bonding

Metals are on the left, and non-metals on the right, they are separated by a staircase line going down from Boron(B) to Astatine(At) Cool. Now you�ve heard of metals, and non-metals. There must be a million differences between these. Including that metals are metallic, and non-metals aren�t. Here are the chemical differences:
Now what is an ionic, or covalent bond? Well, ionic bonds have only one metal element, and non-metal element; for example, NaCl is an ionic bond because it contains only a metal (Na), and a non-metal (Cl), so is Al2O3, two metal aluminum atoms(Al) are joined with three non-metal oxygen atoms(O) to form an ionic bond. Covalent bonds have two or more non-metal elements; for example CO2 is a covalent bond because it has two non-metal elements, carbon(C), and oxygen(O). Now what drives these two atoms together? Electrons!(exciting I know) How? Well remember from the last lesson that atoms try to achieve 0 or 8 electrons in their outer shell by forming bonds with other atoms(Octet Rule), well here is the table from the last lesson.
With ionic bonds, you already know a metal is joined with a non-metal, and that each atom involved either achieves 0 or 8 in their outer shell (Octet Rule), and that the metal gives up electrons while the non-metal takes electrons. Now just how many electrons are given or taken? As many as needed for every atom involved to have 0 or 8 electrons in it�s outer shell. For example, NaCl is an ionic bond, Na has one electron in it�s outer shell, Cl has seven. Na gives the one electron to Cl, so Na can have 0, and Cl can have 8.
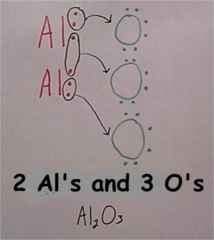
A picture can also be drawn. Now some ionic bonds have more than just one metal atom and one non-metal atom. For Example, Al2O3 has two aluminum atoms and three oxygen atoms, here�s how they form:
A Picture can also be drawn.
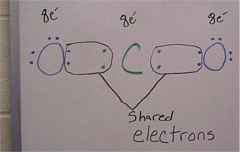
Now, with Covalent bonds, two or more non-metal atoms share a certain amount of electrons until they each have eight in their outer shell. For example, CO2 is a covalent bond that has one carbon atom(C), and two oxygen atoms(O). carbon has four electrons in it�s outer shell, it can share four more electrons, two with the first oxygen atom, two with the other, since each oxygen atom has 6 in it�s outer shell and needs two more to get to eight, they share two of their electrons with the C so all three have eight electons.
 I hope this lesson helps you understand the basic principal of bonding, The next lesson will tell you how to form one of these bonds. Review:
|