|
 |

|

|
|

|
|
Thank Yous
(coming Soon!)
|
|
#27 - Stoichiometry Calculations of |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | | | 1 mole NaF_____ | = .07145 moles NaF |
| | | 41.99 |
| 2 | | | 1 mole LiOH_____ | = .08351 moles LiOH |
| | | 23.949 |
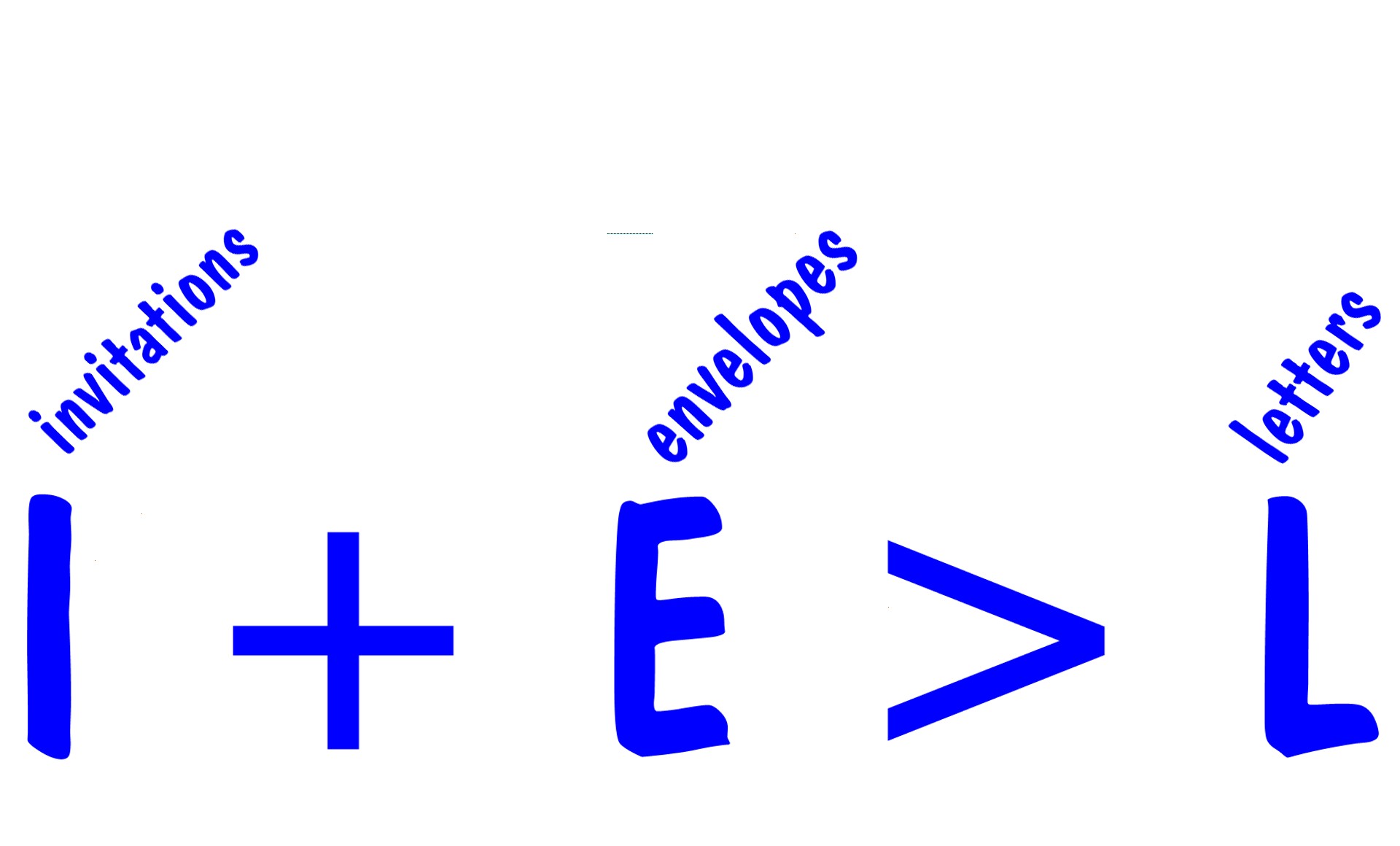
Since there�s less NaF than LiOH, and the mole ratio is 1to1(since the coefficients are both one), NaF is the limiting reactant. The amount of NaF(in moles) determines how much product is made, since NaF will run out before the LiOH just like the envelops will run out before the invitations.
- Use the limiting reactant in figuring out the amount of a product
-So lets find out the amount of product!
| 3 grams NaF | | | 1 mol NaF _________ | | | 1 mol LiF__ | | | 25.9394 g LiF | * | = 1.85 g LiF |
| | | 41.9882 grams NaF | | | 1 mol NaF | | | 1 mol LiF |
The amount of moles does not determine the limiting reactant!
To make things easier, there�s a 5 step process in which you can calculate the limiting reactant and the amount of the product.

|
Here�s the process in action:
Kelly the chemist has 3 grams of Aluminum Nitrate(Al(NO3)3) and 4 grams of Sodium Chloride(NaCl), how much Aluminum Chloride will Kelly make?
1. Make and balance an equation:
Al(NO3)3 + 3NaCl --> 3NaNO3 + AlCl3
2. Get moles of reactants:

|
3 | | | 1 mole Al(NO3)3_____ | = .01818 moles Al(NO3)3 |
| | | 165.01 |
| 4 | | | 1 mole NaC1_____ | = .06845 moles NaCl |
| | | 58.44 |
| .01818 moles Al(NO3)3 | = .01818 |
| .06845 moles NaCl | = .02282 |
4. The reactant with smallest amount from step 3 is the limiting reactant:
.01818 < .02282
Al(NO3)3 is the limiting reactant
5. Use the limiting reactant to find grams of product
| 3 | | | 1 | | | 1 | | | 133.33 grams AlCl3 | =2.4 grams
AlCl3
|
| |
| 165.01 | |
| 1 | |
| 1 | |