|
 |

|

|
|

|
|
Thank Yous
(coming Soon!)
|
|
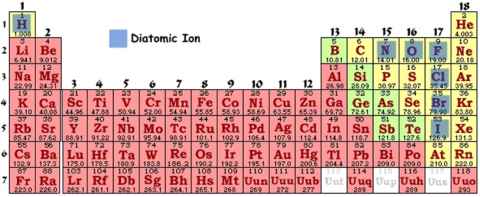
#15 Diatomic ionsThere are some atoms that want to achieve low energy (like stated in the octet rule) so bad that they just pair with another atom of the same kind. These are called diatomic ions. These are just certain non-metals that prefer to be in pairs until they can bond with another element that can satisfy the octet rule even more. There are only seven non-metals that do this:
You really should remember these! Almost all of these elements in supply are diatomic ions. They must be talked about as if they are in diatomic form either before or after reactions if one of these elements might be by itself(not bonded to any other elements) . Each diatomic ion has it�s own, pretty easy to remember, name which is basically the element, plus the phase that element is in (solid, liquid, or gas):
Just look upon it as a "7" on the periodic table, then plus hydrogen:
|