| ANALYSIS OF WATER
In 1783, Lavoisier repeating experiment performed by Priestley and Cavendish, reported
that when inflamable air is burned in ordinary air, in closed vessel, the result was
"water in a very pure state."
In 1785, Lavoisier "decomposed and recombined" the elements in water in a single experiment.
The experiment was performed in front of a large audience and lasted for three days.
He passed water vapor over incandescent iron, to decompose it into hydrogen and oxygen.
The two gases were collected in separate containers and then mixed in a glass balloon and
ignited to form water.
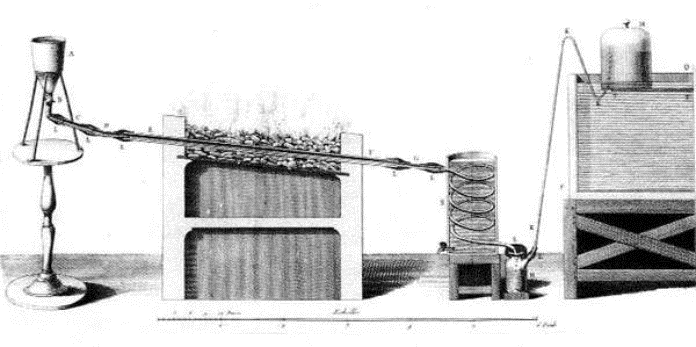
So Lavoisier claimed that water was not an element - but a compound of the two gases oxygen
and inflamable air (gas we now call hydrogen).
|


