|
Synthetic fibers come into existence due to the
result of extensive research done by scientists to increase and improve
upon the supply of naturally occurring animal and plant fibers that have
been used in making cloth and rope.
Generally, synthetic fibers, or man-made fibers, are made by forcing,
usually through the act of thrusting out the fiber forming materials
through holes (called spinnerets) into the air, forming a thread.
| Some common synthetic fibers |
Some specialty synthetic fibers |
| Rayon |
Vinyon |
| Acetate |
Saran |
| Nylon |
Spandex |
| Modacrylic |
Modal |
| Olefin |
Orlon |
| Acrylic |
|
| PLA |
|
 Dyeing
Process of Synthetic Fibers (for helogenious fibers) Dyeing
Process of Synthetic Fibers (for helogenious fibers) |
The process of synthetic fiber (a bundle hair,
comprises halogen containing synthetic fibers) dyeing uses a dyeing
solution containing a dye, a carrier and a solvent for synthetic fiber
hair at 60° to 90° C. The said carrier is used in an amount of
0.05 to 1.2% by weight based on said dyeing solution and said solvent is
used in an amount of 0.05 to 5% by weight based on said dyeing solution.
 Popular
Dyes For Synthetic Fibers Popular
Dyes For Synthetic Fibers |
| Name of fibers |
Name of common dyes that are used |
| Polyester |
Disperse Dyes |
| Acetate (Cellulose acetate) |
Doesn't take dyes ordinarily. It requires cross dyeing. |
| Acrylic |
Disperse dyes |
| Modacrylic |
Fiber Reactive Dyes |
| Modal |
Fiber Reactive Dyes |
| Nylon |
Cationic Dyes, Acid Dyes, Disperse Dyes |
| Orlon |
Disperse Dye |
| Rayon |
Acetate Rayon Dyes |
| Saron |
RIT Dyes |
| Spandex |
Disperse Dyes |
| Vinalon |
Doesn't take Dyes (in normal condition) |
| Vinyon |
Doesn't take Dyes (in normal condition) |
 Definition
of Some Important Dyes Definition
of Some Important Dyes |
RIT Dyes are the chemical dyes which are available in
both a powder dry form as well as in liquid form. The use of the dry
dyes is more preferred over the liquid dyes because the dry dyes store
better for a long period of time, both work equally as well. RIT Dyes
are available at most grocery stores.
Fiber reactive dye is the most permanent of all kind of other dyes bond
with the cellulose or protein molecules. No wonder you can safely wash a
garment that has been dyed in bright fiber reactive colors with white
clothing, a hundred times, without endangering the whites in the least -
even if it is all different bright colors, or even solid black.
Basic dyes are cationic, which means that the colored part has a
positive charge,at the time of dissolving in water. They will bond to
either carboxyl or sulfonic acid groups on a fiber through the formation
of salt links with these anionic groups in the fiber.
 Major
dyes for Synthetic fibers Major
dyes for Synthetic fibers |
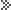 Common
Methods of Dyeing Nylon Common
Methods of Dyeing Nylon |
Using Acid dyes
Although Nylon is a synthetic fiber but due to its some unique
features, it can be dyed like silk or woolen clothes by ACID dyeing. For
this, a typical recipe calls for dissolving dye and salt in adequate
water to cover the material and allow it to move freely in the pot,
adding the material, heat it to a simmer for ten minutes, add some
vinegar and simmer for another ten minutes, then allow to cool gradually
and rinse out. Acid dyes are used for wool, silk, and some synthetics.
Unlike the vat and sulfur dyes, they are water-soluble and can be
applied directly to the fiber.
Using Fiber reactive dyes
Most fiber reactive dyes, specially those which are commonly used to
dye cotton and rayon, cannot react with nylon. But, fiber reactive dyes
can be treated as Acid dyes if some acid like vinegar is added with it
with the necessary heat supply. When used with acid dye recipe, a fiber
reactive dye actually acts as an acid dye.
Using Union dyes
Unions are materials made from two or more different kinds of fibers,
such as cotton and wool or viscose and acetate rayon.
Using Natural dyes
Many natural dyes are acid dyes, which work well on nylon if heated in
a dye bath with an acid, like vinegar or citric acid. Cochineal dyes
nylon a good intense red with a slightly brownish tone, whether
mordanted with alum or not.
However, indigo is considered to perform poorly on all synthetic
fibers, including nylon, because the fibers lack of the internal spaces
found in natural fibers into which the soluble form of the dye may
penetrate and then become trapped upon oxidation.
Using Discharge dyes
The dye in nylon must not be discharged with hydrochloride (ordinary
household chlorine bleach), because the fiber itself, like wool or silk,
will be severely damaged.
|
|