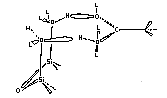
Geminal Conformer

What is Fractal Analysis?
Fractal analysis is a new tool that is being applied to surface science so that we can enhance our ability to work with surfaces. Fractal analysis involves finding the order within a disordered system and then describing the system in terms of non-integral dimensions. The basic concept is illuminated by applying fractal thought to a geography question: "how long is the coast of North Carolina?"
Most people will answer that it is about 300 miles. But this only applies to the macroscopic viewpoint. The actual surface area of the coast of North Carolina could be many different answers. It all depends on the perspective you take when looking at the coast. If you look with the macroscopic view, 300 miles is good enough, but, if you take a microscopic viewpoint, then you see that it is much longer. You have started to look at the nooks and crannies along the boundary.
This same concern is raised in surface chemistry. The basic question is: what is the true surface area? Eric Keightly Rideal once said:
"Since in general, solids possess such irregular surfaces as well as cracks and fissures which may be submicroscopic in size, the estimation of the specific surface is a matter of some difficulty."
This statement illuminates one of the greatest problems ever faced by surface scientists. Since the true surface area depends on what measuring system you are using (microscopic vs. macroscopic), another question has always been: where do you stop the microscopic view of the surface? Until recently, this has been left up to the individual researchers. Fractals are now making this easier to do.
The true surface area is a much greater concern when studying Heterogenous Chemistry as opposed to Homogenous Chemistry. Heterogenous Chemistry studies the interactions and reactions of substances that are in different physical states, i.e. solid-liquid, solid-gas, and liquid-gas. Most chemical research has gone on in homogenous because dealing with it is much easier. However, nature makes things more difficult, most of the natural reactions that take place are in heterogeneous phases.
In heterogeneous chemistry, the geometry of the system is often the main determining factor in the reactibility of the system. The geometry of the system is covered by fractal analysis. The most basic equation for fractal analysis is:
(Number of Yardsticks) (size of the area yardstick) - D / 2
This equation basically delineates the concept of the fact that the area is determined by the relational size of the the measuring system being used.
In surface chemistry we use the equation:
A = N * sigma ( 2 - D) / 2
where A = area
N = Avogadro's number
sigma = cross sectional area
D = fractal exponent
D is the fractal dimension of the surface
and it must be between 2 and 3.
Determination of D
The interaction of any molecule with a surface must depend on the surface structure and the molecule-surface bond. These are the two considerations in developing D: (1) the geometric suitability and (2) the chemical suitability.
The geometric suitability is easy to work with, the fractal method will self determine the suitability of the geometric surface. If the D factor falls between 2 and 3, then the surface is geometrically suitable.
The chemical suitability is more difficult. The major concern in chemical suitability is the heterogeneity of the system. Much of the work with fractals, to date, has been with weak heterogeneous systems. In the weak systems, the geometry factor so outweighs the chemical factor as to render it unimportant. Work has been done to prove the chemical suitability: "the fully hydroxylated surface of silica will hydrogen-bond hydroxyl-bearing adsorbates like alcohols so strongly that the heterogeneities become irrelevant, thus allowing concentrated studies of the geometric factors. However, if the silicas are heated too much, chemisorption occurs at the strained bonds and then the heterogeneity becomes a factor due to the chemistry and geometry becoming inseparable." Work is continuing with weak heterogeneous systems. Work is also now starting to expand into strong heterogeneous systems.
Due to the work done to date, it has been determined that a surface cannot be thought of as simply one surface. We must begin to look at surfaces as a collection of "Effective Surfaces." These effective surfaces must be determined for any specific surface process. For example, the effective surface of silica for a kinetics experiment would be different that the effective surface of silica for an adsorption reaction. The surfaces may be coincidental or they may be different for the different processes, but the effective surface should be determine for the specific reaction.
The actual methods for determining D are varied:
1) Adsorption of small molecules - molecular accessibility analysis
2) Adsorption - particle size analysis
3) Adsorption pore size distribution analysis
4) SAXS
5) SANS
6) Microscopy and image analysis
7) Energy transfer
8) Adsorbed film thickness analysis
9) NMR
10) Fitting to the fractal form of the BET equation
11) Fitting to the fractal form of the FHH equation
The range of available numbers for D is fairly limited. Since the number has to fall between 2 and
3, the fractal analysis is limited to surfaces that have a D between these two numbers. Work has been done on fractals with a D value
above 3 and the method still is applicable, but the uncertainty of its analysis becomes greater. If a value of 2 is found for the D value, then the surface will be absolutely smooth. This was one of the methods used to prove the theoretical work of developing the fractal method. Controlled pore glass and graphite were analyzed for their respective D values. Scientists expected to obtain a number that would be exceptionally close to 2. The actual values found were 2. Since the two compounds have almost perfectly smooth surfaces, this then verified the validity of the fractal method.
Since the value of 2 is smooth, then 3 must be a rough surface. This has been proven in empirical
studies. The closer to 3, the D value is, the rougher the surface. As stated above, values outside of the range of 2-3 start to break the fractal method down. The major determining factor in whether to use the fractal method is the scientist doing the work.
Other work done to prove the fractal method includes the use of microscopes, empirical data, changes of D as the material changed, and comparison of D obtained by multiple methods. The empirical work showed that a great majority of all surfaces give a D value between 2 and 3. The change work was done on carbon being converted to graphite. In this work it was possible to determine the D value at different times and the values were continually moving toward 2, which is the applicable value for graphite.
Use of Fractal Analysis in Surface Chemistry
Fractal analysis is finding many uses in surface chemistry. The analysis is used in adsorption reactions, with both chemisorption and physisorption. It is also find tremendous uses in kinetics. Oftentimes the surface area will greatly increase the speed of a reaction, or it may greatly decrease the speed of a reaction.
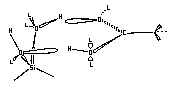
A major use for the fractal analysis method evaluating the molecular conformations in the adsorbed state. An example of this is the determination of the adsorbed conformation of n-alkanoic acids on a silica-toluene interface. In this example, the fractal value of the silica was determined by other means, thus allowing us to backcalculate the sigma value and thus determine the referred conformation for adsorption. The two conformers are:

Geminal Conformer

The conformer favored was the geminal conformer. It has a smaller cross sectional area and the fractal
analysis showed that the smaller molecule was adsorbed.
In determining the conformer adsorbed, the surface of the silica-toluene interface was shown to have a similar effective surface for all molecule -surface interactions. The geminal conformer is the horizontal molecule and previous work was done suggesting that this would be the adsorbed molecule. The fractal method also gave excellent agreement on the cross sectional area of these molecules.
Another use of fractal analysis is in the determination of a cross sectional area of a polymer. This analysis is done in the same manner that the conformational analysis was done for the alkanoic acid.
Fractal Analysis is a new method for surface science. It has many applications, much of them due to now being able to study the natural chemical processes that surround us. Finally, we are able to mathematically describe the actual true surface area for many surfaces.
Bibliography
E. K. Rideal, An Introduction to Surface Chemistry, Cambridge University Press, London, 1930, pp. 175-176.
Avnir, D., Farin, D., Pfeifer, P., "A Discussion of some aspects of Surface Fractality and of its Determination", New Journal of Chemistry, 16, p. 442.
Avnir, D., Farin, D., Pfeifer, P., "A Discussion of some aspects of Surface Fractality and of its Determination", New Journal of Chemistry, 16, p. 441.

| Return to Sending Page |
| Duane A. Floyd's Science Home Page |
| Quick Links To | Duane's Major Pages | ||
| |
|||
| Home | Professional | Humanities | Books |
Page is � 1999 by Duane A. Floyd