 |
| By, Cheri McGough |
| Symbol: Xe Atomic Number: 54 Atomic Mass: 131.29 amu Melting point: -111.9 degrees C Boiling point: -108.1 degrees C Number of Protons/ Electrons: 54 Number of Neutrons: 77 Classification: Noble Gas Crystal Structure: Cubic Density at 293 K: 5.8971 g/cm cubed Color: Colorless Gas |
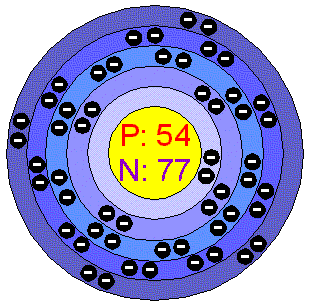 |
| Atomic Structure: |
| Number of Energy Levels: 5 ~First Energy Level:2 ~Second Energy Level: 8 ~Third Energy Level: 18 ~Fourth Energy Level: 18 ~Fifth Energy Level: 8 |
| Facts: Date of Discovery: 1898 Discoverer: Sir William Ramsay Name Origin: From the Greek word xenon (stranger) Uses:power lamps, bubble chambers Obtained From: liquid air |
 |
| More essentials: ~group number: 18 ~period number: 5 ~Block: p-block ~standard state: gas at 298 K ~classification: Non-metallic ~xenon is a "noble" or "inert" gas present in the atmosphere to a small extent ~It can be highly explosive xenon trioxide ~*Metallic xenon is produced by applying several hundred kilobars of pressure. Xenon in a vacuum tube produces a blue glow when excited by an electrical discharge and finds use in strobe lamps. It is an odorless, colorless, inert gas.*~ |
| Isolation of xenon: Xenon is present to a small extent in the atmosphere (less than one ppm by volume) and is obtained as a byproduct fromt he liquefaction and separation of air. This would not normally be carried out in the laboratory and xenon is available commercially in cylinders in high pressure. |