Autoionization Of Water
![]()
The autoionization of water is an equilibrium reaction that is written:
H2O(l) ![]() H+(aq) + OH-(aq)
H+(aq) + OH-(aq)
The equilibrium constant expression for this reaction is given by:
Keq = Kw = [H+] [OH-] = 1.0 x 10-14
The value for Kw is for room temperature, 25 °C, and 1.0 atm of pressure.
The equilibrium constant expression applies not only to pure (distilled) water but to any aqueous solution. It can be used to calculate either [H+] or [OH-] provided one of them is known.
When [H+] = [OH-], the solution is neutral, when the [H+] > [OH-] the solution is acidic, and when [H+] < [OH-] the solution is basic. [H+] and [OH-] are inversely proportional meaning their product must always equal the constant, 1.0 x 10-14, at 25 °C, and 1.0 atm of pressure.
The degree of ionization and the value of Kw varies with temperature as shown in the table below.
Temp (°C) |
1/Temp (x 10-3 K-1) |
Kw |
ln Kw |
pKw |
|---|---|---|---|---|
| 0 |
3.66 |
1.14 x 10-15 |
-34.41 |
14.9435 |
| 5 |
3.60 |
1.85 x 10-15 |
-33.92 |
14.7338 |
| 10 |
3.53 |
2.92 x 10-15 |
-33.47 |
14.5346 |
| 15 |
3.47 |
4.53 x 10-15 |
-33.03 |
14.3436 |
| 20 |
3.41 |
6.81 x 10-15 |
-32.62 |
14.1669 |
| 25 |
3.36 |
1.01 x 10-14 |
-32.23 |
13.9965 |
| 30 |
3.30 |
1.47 x 10-14 |
-31.85 |
13.8330 |
| 35 |
3.25 |
2.09 x 10-14 |
-31.50 |
13.6801 |
| 40 |
3.19 |
2.92 x 10-14 |
-31.16 |
13.5348 |
| 45 |
3.14 |
4.02 x 10-14 |
-30.84 |
13.3960 |
| 50 |
3.10 |
5.47 x 10-14 |
-30.54 |
13.2617 |
The form of the van't Hoff equation which is valid for small temperature ranges is given by:
ln K = -ΔHrxn°/RT + ΔSrxn°/R
Comparing this equation to y = mx + b, suggests that a plot of ln Kw vs 1/T will be a straight line with a slope of -ΔHrxn°/R and the term ΔSrxn°/R is the y-intercept as shown in the graph below.
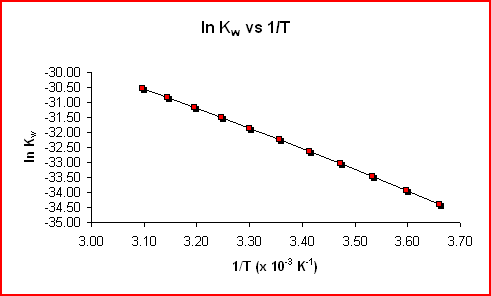 |
Applying the van't Hoff equation and ignoring the y-intercept gives:
ln (K2/K1) = (-ΔHrxn°/R)(1/T2 - 1/T1)
Substituting the values corresponding to 50 °C and 0 °C gives:
ln ((5.47 x 10-14)/(1.14 x 10-15)) = (-ΔHrxn°/8.31 J/mol•K)(3.10 x 10-3 K - 3.66 x 10-3 K)
ΔHrxn° = 57.43 kJ/mol
The graph below shows the line of best fit (linear regression) for the data above.
 |
The graph of pKw vs 1/T shown below also gives the same result. The slope, m, of the graph below is given by:
m = ΔpKw/Δ(1/T) = pKw2 - pKw1/(1/T2 - 1/T1)
The slope of the graph, m, also equals:
m = ΔHrxn°/(2.3026•R)
Equating these two expressions for m and substituting values yields:
(13.262 - 14.944)/(323-1 K-1 - 273-1 K-1) = ΔHrxn°/(2.3026•8.31 J/mol•K•1 kJ/103 J)
ΔHrxn° = 56.76 kJ/mol
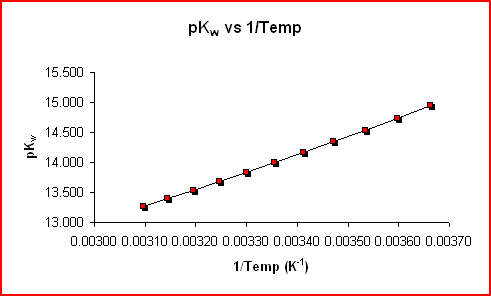 |
The graph below shows the line of best fit (linear regression) for the data above.
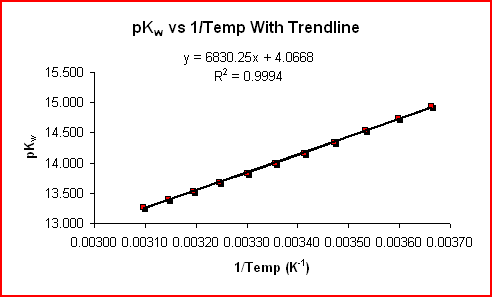 |
The value for ΔHrxn° obtained from the graph compares favorably to ΔHrxn° when computed using enthalpies.
H2O(l) ![]() H+(aq) + OH-(aq)
H+(aq) + OH-(aq)
ΔHrxn° = ΣnΔHf°(products) - ΣmΔHf°(reactants)
ΔHrxn° = [ΔHf°(H+(aq)) + ΔHf°(OH-(aq))] - [ΔHf°(H2O)]
ΔHrxn° = (0 + 1 mol OH- x -230.0 kJ/mol OH-) - (1 mol H2O x -285.83 kJ/mol H2O) = 55.8 kJ