by, Sithamalli K. Balasubramanian - Ph.D.
Our
work on Boron is complete but the documentation and preparation
of the paper are not. Hence the following comments are offered
to show the scope of the model. The presentation should be considered
tentative and is not ready for publication. 11B is assigned
structure
(1) without the apex of the tetrahedron. This small change
leads to a big change in the bonding propensity of the element.
There
are at least three bonding patterns for boron in the pentavalent
state. These are given in (2a, 2b, 2c). (The trivalent state
poses no problem.)
Boron is pentavalent with elements on the left side of the periodic table and trivalent on the right side. The pattern 2a has four bonds pointing downwards whilst the last one points upwards at an angle to the vertical. This is different from the square carbon atom because the distribution of the four downward bonds is rectangular.
Pattern 2a is present in crystalline boron which has boron icosahedra linked to neighbouring icosahedra at an angle . This is a unique structure for any element. If boron has all the five bonds pointing in one direction each icosahedron would be self-contained and would have no valence for further links. Like the inert gases elemental boron would be self-contained with each molecule having an independent existence with no tendency for intermolecular linkages. In reality boron is one of the hardest substances. Each boron atom is linked to four boron atoms in the icosahedron and has one bond for inter-icosahedral linkage as shown in line drawing (3).
The bonding pattern 2a is also to be found in some cyclic boron hydrides.
Pattern 2b is found in diborane, (4). 2b is also found for terminal boron atoms in some linear hydrides. Diborane has doubly bound boron atoms with one hydrogen attached to each boron at the central area of the B=B.
Pattern 2c is found in hydrides. ...top
Boron is pentavalent with elements on the left side of the periodic table and trivalent on the right side. The pattern 2a has four bonds pointing downwards whilst the last one points upwards at an angle to the vertical. This is different from the square carbon atom because the distribution of the four downward bonds is rectangular.
Pattern 2a is present in crystalline boron which has boron icosahedra linked to neighbouring icosahedra at an angle . This is a unique structure for any element. If boron has all the five bonds pointing in one direction each icosahedron would be self-contained and would have no valence for further links. Like the inert gases elemental boron would be self-contained with each molecule having an independent existence with no tendency for intermolecular linkages. In reality boron is one of the hardest substances. Each boron atom is linked to four boron atoms in the icosahedron and has one bond for inter-icosahedral linkage as shown in line drawing (3).
The bonding pattern 2a is also to be found in some cyclic boron hydrides.
Pattern 2b is found in diborane, (4). 2b is also found for terminal boron atoms in some linear hydrides. Diborane has doubly bound boron atoms with one hydrogen attached to each boron at the central area of the B=B.
Pattern 2c is found in hydrides. ...top

Fig. 1 - Boron Atom

Fig. 2a - Bonding pattern of boron

Fig. 2b, 2c - Bonding patterns of boron
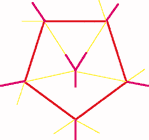
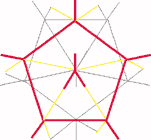
Fig. 3a, 3b - Bonding pattern in crystalline Boron.

Fig. 4 - Diborane structure. There is one hydrogen above and one below the plane of the molecule.