Nuclear
Fission
Back
to Physics World
Back to Special Relativity
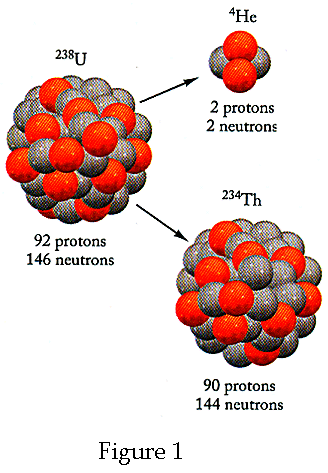
The relationship between mass and energy can be understood more readily by considering the energy released in nuclear fission. Consider the spontaneous fission show below, which takes place in frame S. Let the original Uranium nucleus be at rest prior to fission and therefore the frame S is the zero-momentum frame. (Note: The term mass when used unqualified, means inertial (aka relativistic) mass). The nuclear equation for this reaction is (See Figure 1 below)
![]()

Note:
1 u º 931.48MeV/c2
m0
º Initial mass of the system = proper mass of Uranium
isotope.
m0n
= Proper mass of a the nth particle.
mn
= Mass of a the nth particle.
U
= Potential energy associated with fission fragments in the initial bound state.
K º Final total kinetic
energy of fission products.
![]()
U/c2
is the mass associated with the potential energy, U, of the bound state
of the initial nucleus and
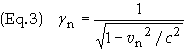
The Q
of a the reaction in Eq. (1) is defined by
![]()
Thus
the energy given off in this fission process equals the change in potential of
the alpha particle inside the thorium nucleus. Substituting in the values in the
table above gives
![]()
The
energy of a free moving particle is the sum of its kinetic energy and its rest
energy. Therefore

As calculated above, Q = U = K. The change in potential energy results in the kinetic energy of the fission products. Since the Thorium nucleus is much greater than the alpha particle (i.e. 4He = Helium isotope) then the total kinetic energy remains in the alpha particle. Therefore the Q of the reaction is the total kinetic energy of the fission fragments.
(Note:
If any photons are produced during nuclear fission then they are treated as
particles with zero rest mass. The energy is treated as kinetic energy).
References:
* CRC Handbook of Chemistry and Physics – 86th Ed., David R. Lide (Editor-in-Chief), Edition 2005-2006.

Back
to Special Relativity
Back to Physics World