Study of chemistry of life
Atoms: the smallest unit of matterneutrons: neutral subunit of atomic nucleusprotons: positively charged subunit of atomic nucleus
electrons: negatively charged particles surrounding the nucleus
Elements: group of one type of atom
Common elements in the earth’s crust:Oxygen: 47%Silicon: 28%
Aluminum: 8%
Iron: 5%
Other elements: 12%
Common elements in dry air
Nitrogen: 78%Oxygen: 21%
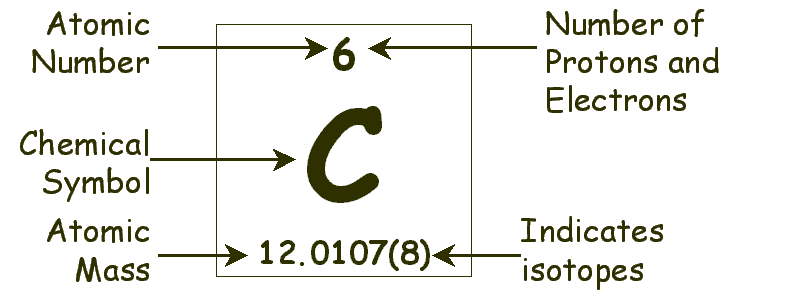
The Periodic Table

Covalent Bonds: non-metal elements share electrons to complete outer shells.Ionic Bonds: metal and non-metal elements lose/ gain electrons to complete outer shells.
Hydrogen Bonds: slight attraction between slightly charged molecules.
Common Elements
Ca: CalciumSn: Tin
Au: Gold
Ag: Silver
Cu: Copper
Fe: Iron
Hg: Mercury
Na: Sodium
Cl: Chlorine
Six common elements in living organisms:
C: CarbonH: Hydrogen
O: Oxygen
N: Nitrogen
P: Phosphorus
S: Sulfur
Biochemistry: Organic Compounds
Organic Compounds: compounds made and used by living organisms.Must contain carbon and hydrogen.
95% of living matter is made of C, H, O, and N
99% of living matter is made of C, H, O, N, P, and S
Carbohydrates
Elements: carbon, hydrogen, and oxygen.Building Blocks: monosaccharides
Examples
glucose: blood sugarsucrose: table sugar
starch: plant storage molecule
glycogen: animal starch
cellulose: plant building molecule
Function: energy, structure
Lipids
Elements: carbon, hydrogen, and oxygen.Building Blocks: fatty acids and glycerol
Examples
saturated fat: solid at room temperatureunsaturated fat: liquid at room temperature
cholesterol
waxes
steroids
Function: energy storage, insulation
Proteins
Elements: carbon, hydrogen, oxygen, nitrogen, and sulfur.Building Blocks: amino acids
Examples
musclehair
nails
feathers
Function: enzymes in biochemical reactions; structural materials
Nucleic Acids
Elements: carbon, hydrogen, oxygen, nitrogen, and phosphorus.Building Blocks: nucleotides
Examples
DNARNA
Function: store genetic information