Nuclear Decay 
In a sample of radioactive nuclei, each nuclide has a certain probability of undergoing radioactive decay. The rate of decay is the negative of the change in the number of nuclides per unit time as shown below:
Rate = -ΔN/Δt
The decay rate is directly proportional to the number of nuclides as shown below:
Rate = -ΔN/Δt α N
To change the above expression into a useful equation, a proportionality constant k is used as given below:
Rate = -ΔN/Δt = kN
The equation above is the rate law for a first-order process. The integrated first-order rate law is given by:
ln(N/N0) = -kt (1)
N0 represents the original number of nuclei at t = 0 and N represents the number remaining at time t.
The half-life is defined as the time necessary for one half of a radioactive sample to undergo transmutation (decay) into new elements. Different isotopes have different decay rates. To determine the expression for the half-life (t1/2), N = N0/2 is substituted into equation (1) which yields:
ln(1/2
N0/N0) = -kt1/2 (2)
Equation 2 can be simplified further as shown below:
ln(1/2) = -kt1/2
ln(1) - ln(2) = -kt1/2
t1/2 = -ln(2)/-k = 0.693/k
where k is the rate (decay) constant.
Mass values can be substituted for the N values because the mass will be proportional to the number of nuclei.
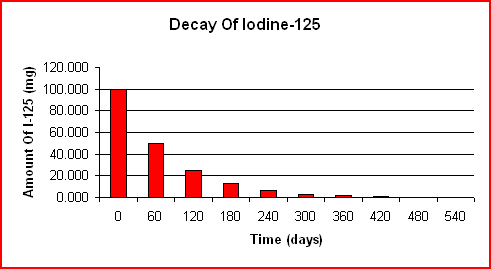
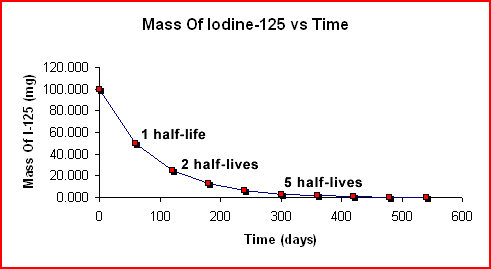
The decay of a 100.000-g sample of I-125 over time is shown below. Note that the half-life is a constant 60 days in both graphs.