Liver and biliary tract
The importance of
determination of
alveolo-arterial
gradient in patients with liver cirrhosis
1Đorđe
Ćulafić,
2Predrag
Rebić
1.
Institute of Digestive Diseases,
Clinical
Centre of Serbia Belgrade
2.
Institute of Pulmonary Diseases
and
Tuberculosis,
Clinical
Centre of Serbia Belgrade
ABSTRACT
It is demonstrated that
alveolo-arterial gradient ≥ 2 kPa with or without hypoxemia indicates
intrapulmonary vascular dilatation. In order to assess the importance of
determination of alveolo-arterial gradient in patients with liver cirrhosis, we
analyzed results of 70 patients with liver cirrhosis. The oxygen pressure in
the alveolar gas was calculated on the basis of the measured atmospheric
pressure and carbon-dioxide in the exhaled air, while Pa,O2 was measured
directly in the arterial blood. Orthodeoxia was confirmed in 10 (14.3%)
hypoxemic and 4 (5.7 %) normoxemic patients. In our series, 30 (42.8%) patients
had alveolo-arterial gradient over 2 kPa. All patients with orthodeoxia had
higher alveolo-arterial gradient, whose mean value in the supine position was
4.03 ± 2.36 kPa while it was 5.73 ± 2.65 kPa in the sitting position. Among the
patients with restrictive ventilatory disorders caused by ascites and without
orthodeoxia, the mean value of P(A-a)O2 was
2.77 ± 1.12 kPa in the supine position while it was 1.80 ± 1.04 kPa in the
sitting position. The data suggests that orthodeoxia and higher
alveolo-arterial gradient are sensitive functional parameters in the diagnosis
of intrapulmonary vascular dilatation, which represent, the major pathogenic
mechanism in the development of severe respiratory disorders in liver
cirrhosis.
INTRODUCTION
Celiac disease
represents by definition a hypersensitivity of In
patients with liver cirrhosis and prominent hypoxemia, the IPVD and
right-to-left shunts represent the major pathogenic mechanism in the
development of severe respiratory disorders (1–3).
The most common
pulmonary symptom in IPVD is dyspnea, affecting almost 40% of patients. If
questioned, up to 70% of candidates for liver transplantation complain of
dyspnea (4). Another typical manifestation of IPVD in cirrhosis is platypnea,
specifying that dyspnea is more pronounced in the upright and sitting position
than in a recumbent one (5). Orthodeoxia is another clinical sing in IPVD,
representing the decrease of partial oxygen pressure more then 10% when patient
is changing the position from a recumbent to a sitting one.
Hypoxemia with
partial oxygen pressure (Pa,O2) less than 8 kPa without any other cardiorespiratory
diseases suggests the presence of IPVD (6,7).
The determination
of P(A-a)O2, is the simplest method of assessment the
ratio of ventilation and perfusion of the lungs. It is demonstrated that P(A-a)O2 ≥ 2 kPa with or without hypoxemia indicates the IPVD
(8).
PATIENTS AND METHODS
The prospective
study analyzed results of 70 patients with liver cirrhosis which were treated
at the Institute of Digestive Diseases and Institute
of Pulmonary Diseases, Clinical Centre
of Serbia.. Gas analyses were carried out using Blood Gas Manager IL
1312 equipment, while predicted values for the partial oxygen pressure in blood
were calculated using Sorbini equation (Pa,O2 =103.5 - 0.42 x years) (9). Arterial blood gases analysis
were performed in both supine and sitting positions while exposed to the room
air and after 15 minutes of breathing of hyperoxic mixture using a direct
method, which includes arterial blood sample with a heparinized syringe.
Spirometric
measurements in our study were performed by open spirometric system with
pneumotachograph (Pneumoscreen II spirometer) for determination: vital capacity
(VC), forced vital capacity (FVC) and forced expiratory volume during the first
second (FEV1). Body plethysmograph (Bodyscreen II) was used for measurement of
total lung capacity (TLC), thoracic gas volume (TGV), residual volume (RV), and
air volume resistance in the airways (RaW).
The
alveolo-arterial gradient of oxygen pressure (P(A-a)O2=PA,O2-Pa,O2) was calculated in the way usually used in
practice. The oxygen pressure in the alveolar gas is calculated on the basis of
the measured atmospheric pressure and carbon-dioxide in the exhaled air (PA,O2= Fio2 x (Pb – PaH2O) – PA,CO2 (Fio2 + 1 – Fio2 / Ra) ( Fio2-fractional
inspiratory concentration of oxygen; Ra-respiratory quotient), while Pa,O2 is measured directly in the arterial blood
(10).
Given to the
variations of respiration changes in cirrhotics while assuming different body
positions, arterial blood gas changes occurred, the change of P(A-a)O2 for at least 0.66 kPa is a sign of
significant alteration of IPVD with the assumption of different body positions
(11).
Pulmonary
function tests were used to determine the transfer factor (TL,CO) and transfer coefficient (KCO = TL,CO divided by the effective alveolar volume), as the indicators
of alveolo-capillary diffusion state. Diffusion parameters were measured by
carbon monoxide, using the one-inspiration method, with Transferscreen machine.
Lower normal limit of transfer factor was determined by mathematics in the way
that the expected values of TL,CO were subtracted by SD of 1.64 (SD of 1.42 for males, SD of
1.17 for females). The expected values of TL,CO were calculated on the basis of valid
standards (in males, expected TL,CO = 11.11 x height in meters -0.066 x years - 6.03; in females,
expected TL,CO =
8.18 x height in meters - 0.049 x years - 2.74) (11).
Restrictive
ventilatory disorders were defined on the basis of spirometric parameters: VC,
index 100 x FEV1/VC (Tiffeneau) and TLC. Lower normal limit was determined as
the expected value – 1.64 SD (in males, SD for FVC = 0.61, SD for TLC = 0.70,
and for Tiffeneau SD
= 7.17; in females, SD for FVC = 0.43, SD for TLC = 0.60, and for Tiffeneau SD
= 6.51) (12).
Statistical
analyses were performed using χ2 and t-test.
RESULTS
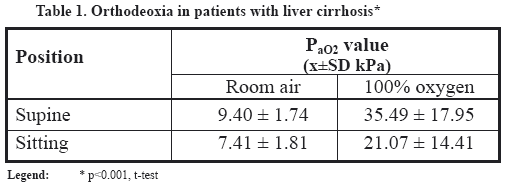
Orthodeoxia was
confirmed in 10 (14.3%) hypoxemic and 4 (5.7 %) normoxemic patients. When room
air was breathed, the average Pa,O2 value in a supine position was 9.40 ± 1.74
kPa, while in a sitting position the average Pa,O2 value was 7.41 ± 1.81 kPa. When 100% oxygen was breathed, the
average Pa,O2 value in supine position was 35.49 ± 17.95 kPa, while in the
sitting position the average Pa,O2 value was 21.07 ± 14.41 kPa (Table 1).
In our series, 30
(42.8%) patients had P(A-a)O2 over 2 kPa. All patients with orthodeoxia
had higher P(A-a)O2, whose mean value in the supine position
was 4.03 ± 2.36 kPa while it was 5.73 ± 2.65 kPa in the sitting position. The patients
with orthodeoxia and higher P(A-a)O2 had IPVD. The patients with orthodeoxia and
normoxemia also had higher P(A-a)O2, ranging from 2.1 to 2.37 kPa and
presented subclinical IPVD.
The diagnosis of
ascites was made in 43 (61%) patients. Restrictive ventilatory disorders and
higher P(A-a)O2 were diagnosed in 16 (37.2%) patients with
ascites. Mean value of restriction parameters was: VC 73.0 ± 14.4, TLC 83.7 ±
13.5, and Tiffeneau index 78.3 ± 3.37.
Among the
patients with restrictive ventilatory disorders and without orthodeoxia, the
mean value of P(A-a)O2 was 2.77 ± 1.12 kPa in the supine position
while it was 1.80 ± 1.04 kPa in the sitting position. Comparing patients with
and without orthodeoxia, the significant difference in P(A-a)O2 (p=0.001, t-test) was found, especially in the sitting
position (Table 2).

Lower transfer
factor (TL,CO) was found in 38 (54%) patients, while lower transfer
coefficient (KCO) was
noted in 46 (66%) patients. The mean value of transfer factor was 6.24
(65.72%). All patients with IPVD and subclinical IPVD manifested reduced values
of transfer factor and transfer coefficient.
Higher P(A-a)O2 and normal values of transfer factor were recorded in 11
(16%) (patients with restrictive ventilatory
disorders), while the increased P(A-a)O2 and lower transfer factor were found in 14 (20%) patients
(patients with IPVD). Lower transfer factor and normal P(A-a)O2 were recorded in 17 (24%) patients. No statistical
significance between the increase of P(A-a)O2 and diffusion impairment was found (χ2
– test, p=0.62) (Table 3).

DISCUSSION
In their study of
26 patients with liver cirrhosis, Bashour and Cochran (1966) obtained the mean
gradient value of 5.97 kPa (13).
In the group of
patients who were candidates for portocaval shunt, Naeije et al (1985) measured
the mean value of P(A-a)O2 of 4.58 kPa (14).
In 1991, Hourani
and associates reported the increased gradient of oxygen pressure in 45% of
patients who were candidates for liver transplantation, and the mean value of
gradient was 4.89 kPa (4).
Stressing the
significance of this functional disorder, Fahy and associates (1992) stated
that 69% of patients, being the candidates for liver transplantation, had
higher P(A-a)O2 (15).
In 1998, Behera et
al found hypoxemia with the increased P(A-a)O2 in 26.7% of patients with liver cirrhosis.
Better findings were manifested in patients with extrahepatic obstruction of
portal vein (16).
In Fallon’ cohort
of 207 consecutive liver transplant candidates who underwent arterial blood gas
screening, 66% had P(A-a)O2 higher than 2 kPa (17).
In our series, 30
(42.8%) patients had P(A-a)O2 over 2 kPa. The increased P(A-a)O2 (5.73 kPa ± 2.65), particularly in the sitting position, was
recorded in all patients with orthodeoxia, which indicates the IPVD.
Frequently, the
hyperventilation of patients with liver cirrhosis may increase P(A-a)O2 values even in the case of normoxemia. Therefore, P(A-a)O2 is sensitive parameter of pulmonary gas exchange disorder.
High diagnostic significance of these tests has been corroborated by findings
of the unchanged basal partial oxygen pressure in the presence of subclinical
IPVD, in spite of the increase of P(A-a)O2 (18).
Subclinical IPVD,
as specific entity, present the transitory phase in the development of
hepatopulmonary syndrome (HPS). In spite of IPVD and the increased P(A-a)O2, the partial arterial oxygen pressure may stay unchanged for
a long period of time. This is caused by alveolar hyperventilation,
hyperdynamic circulation, and an increase of cardiac output characteristic for
cirrhotic (19,20).
Subclinical lung
vasculature vasodilatation, with concomitant normoxemia may be found in the
early phases of liver cirrhosis. With the progression of pulmonary changes, the
hypoxemia becomes increasingly present. During the course of the disease, the
impairment of oxygenation may be intensified even without the aggravation of
liver function (21).
In our series,
all four patients with subclinical IPVD were normoxemic despite increased P(A-a)O2, ranging from 2.1 to 2.37 kPa.
Among the
patients with restrictive ventilatory disorders caused by ascites and without
orthodeoxia, the mean value of P(A-a)O2 was 2.77 ± 1.12 kPa in the supine position
while it was 1.80 ± 1.04 kPa in the sitting position.
Patients with
abundant ascites, in standing position, usually have normal results of
respiratory gasses of arterial blood, while partial insufficiency of
respiration is manifested in recumbent position. Early closing of the airways
makes the large areas in lower lung regions not to participate in ventilation
while having maintained perfusion, and, accordingly, the ventilation-perfusion
ratio is substantially worse. Perfusion of non-ventilated regions causes the
situation that a large quantity of blood flows through lungs without being
oxygenated (22).
Chang and
associates (1997) published the results of their study where the effect of
ascites to pulmonary function was monitored in two groups of patients, by
comparison of therapeutically effects of paracentesis and diuretics. In
distinction from patients treated by paracentesis, the patients administering
diuretic therapy manifested significant improvement of gas exchange, along with
PaO2
increase and large
reduction of P(A-a)O2.
The improvement of oxygenation following the diuretics suggests that, other
than mechanical effect of ascites, the interstitial pulmonary edema and fluid
retention contribute additionally to more difficult gas exchange (23).
Studies using 100%
oxygen are also commonly used in patients with documented or suspected IPVD. On
the basis of response to exposure to 100% oxygen, two types of vascular
abnormalities in patients with hepatocellular insufficiency have been
described. Type-1 is characterized by diffuse vascular dilatation at
precapillary level, while, type-2 is defined by true anatomic pulmonary
arterio-venous shunting. The application of 100% oxygen leads to significant
increase of partial oxygen pressure in the type-1, while the effect is minimal
in the type-2 (7).
The incomplete
response with partial correction of hypoxemia was obtained in some patients
following the inspiration of 100% O2. The impairment was recognized as the
result of IPVD, what also contributed to shunt fraction and it was designated
as diffusion-perfusion defect or alveolo-capillary oxygen disequilibrium (24,25).
In 1991, Hourani
and associates described that, out of 116 patients with liver cirrhosis, 60
(52%) cases manifested reduced diffusion capacity. The increased capillary
plasmatic volume and alveolar capillary dilatation give rise to the increase of
diffusion distance, representing the basic mechanism of the impaired carbon
monoxide and oxygen transfer in liver diseases. The reduction of diffusion
capacity in regularly oxygenated cases may be also explained by IPVD, and
subclinical intrapulmonary arterio-venous shunts, respectively. It was
significant that 40% patients in this study, who manifested diffusion
impairment, were not found to have the increase of P(A-a)O2. The authors consider that TL,CO may be more sensitive indicator of minor
IPVD than the increase of P(A-a)O2 (4).
Krowka and
Cortese, reported, in 1994, that in isolated reduction of TL,CO commencement together with the IPVD, other
pathogenetic mechanisms were taking part, such as: diffuse interstitium
pulmonary diseases, causing no restrictive disorders in the early phase, flow
through non-ventilated alveoli, ventilatory-perfusion imbalance and/or other
pulmonary vascular conditions (7).
In our study,
lower transfer factor and normal P(A-a)O2 were noted in 17 (24%) patients. Higher P(A-a)O2 and normal transfer factor were found in 11(16%) patients,
while the increased P(A-a)O2 and
decreased transfer factor were recorded in 14 (20%) patients (cases with IPVD).
There was no significant difference in comparison of these two groups,
suggesting that the isolated reduction of TL,CO could not be explained only by IPVD.
Orthodeoxia and
higher P(A-a)O2 are sensitive functional parameter in
diagnostic IPVD, which represent, the major pathogenic mechanism in the
development of severe respiratory disorders in liver cirrhosis.
1. Edell ES, Cortese DA, Krowka MJ,
Rehder K. Severe hypoxemia and liver disease. Am Rev Respir
Dis 1989; 140:1631–5.
2. Andrivet P, Cadranel J, Housset B,
Herigault R, Harf A, Adnot S. Mechanisms of impaired arterial oxygenation in
patients with liver cirrhosis and severe respiratory insufficiency. Effects of mdomethacin. Chest 1993;
103:500 –7.
3. Robert V, Chabot F, Vial B, et al. . Hepatopulmonary syndrome
physiopathology of impaired gas exchange. Rev Mai Respir 1999; 16:769
–79.
4. Hourani JM, Bellamy PE,
Tashkin DP, Batra P, Simmons MS. Pulmonary dysfunction in advanced liver
disease: frequent occurrence of an abnormal diffusing capacity. Am J Med 1991; 90:693 – 700.
5. Robin ED, Laman D, Horn BR,
Theodore J. Platypnea related to ortodeoxia caused by true vascular lung
shunts. N Engl J Med 1976; 294:941 –3.
6. Rodriguez-Roisin R. The hepatopulmonary
syndrome: new name, old complexities.Thorax 1992; 47:897– 902.
7. Krowka MJ,
Cortese DA.
Hepatopulmonary syndrome: Current concepts in diagnostic and therapeutic
considerations. Chest 1994; 105:1528 –37.
8. Rodriguez-Roisin R. The
hepatopulmonary syndrome: new name, old complexities. Thorax
1992; 47:897 – 902.
9. Sorbini CA, Grassi V, Solinas E,
Muiesan G. Arterial oxygen tension in relation to age in healthy subjects. Respiration 1968; 25:3 – 13.
10. Rebic P. Merenje odnosa
ventilacije i perfuzije u plucima. U: Sekulic S (urednik) Plućne bolesti.
Elit Medica. Beograd 2000:79 – 81.
11. Cotes JE: Standardisation of the
measurement of transfer factor (diffusing capacity).The European Respiratory
Journal 1993; 6:5 – 40.
12. Quanjer PH. Lung volumes and
forced ventilatory flows. The European Respiratory Journal
1993; 6:5- 40.
13. Bashour A, Cochran P.
Alveolar-arterial oxygen tension gradients in cirrhosis of the liver. Further evidence of existing pulmonary arteriovenous shunting.
Am Heart J 1966; 71:734 – 40.
14. Naeije R, Melot C, Hallemans R,
Mols P, Lejune P. Pulmonary hemodynamics in liver cirrhosis. Seminars
in Respiratory Medicine 1985; 7:164 –70.
15. Fahy JV, Kerr KM, Lake JR, Gold
WM. Pulmonary function before and after liver transplantation (abstract). Am
Rev Respir Dis 1992; 143:A303.
16. Behera D, Singh M, Chawla Y,
Dilawari JB. Pulmonary function abnormality in patients with
portal hypertension with or without chronic liver disease. Indian J Chest Dis Allied Sci 1998; 40:33 –9.
17. Fallon MB, Abrams GA. Pulmonary
dysfunction in chronic liver disease. Hepatology 2000; 32:859
–65.
18. Lange PA, Stoller JK. The hepatopulmonary syndrome. Ann Int Med
1995; 22:521 – 9.
19. Agusti AG, Roca J,
Rodriguez-Roisin R. Mechanisms of gas exchange impairment in patients with
liver cirrhosis. Clin Chest Med 1996; 7:49 – 66.
20. Miki K, Shinohara T, Ogushi F, et
al. Hepatopulmonary syndrome-discussion of cardiopulmonary parameters. Med
Invest 2000; 47:164 – 9.
21. Mimidis KP, Karatza C,
Spiropoulos KV, et al. Prevalence of intrapulmonary vascular dilatations in
normoxemic patients with early liver cirrhosis. Scand J Gastroentrol 1998; 33:
988 –92.
22. Gupta D, Lalrothuama S, Agrawal
PN, et al. Pulmonary function changes after large volumen paracentesis.Trop
Gastroentrol 2000; 21:68-70.
23. Chang SC, Chang HI, Juh F, Shiao
GM, Wang SS, Lee SD. Therapeutic effects of diuretic and paracentesis on lung
function in patients with non-alcoholic cirrhosis and tense ascites. J Hepatol 1997; 26:833-8.
24. Davis HH, Schwartz DJ, Lefrak SS, Susam N,
Schainker BA. Alveolar-capillary oxygen disequilibrium in
hepatic cirrhosis. Chest 1978; 73:507 –11.
25. Krowka MJ,
Cortese DA.
Severe hypoxaemia associated with liver disease: Mayo Clinic experience and the
experimental use of almitrine bismesylate. Mayo Clini Proc 1987; 62:164 –73.
20. Swinson CM, Slaavin G, Coles EC,
Booth CC. Celiac disease and malignancy. Lancet 1983; 1:
111-13.
21. Ferguson A. Kingstone K. Celiac
disease and malignancies. Acta Paediatr 1996, 412(suppl): 78-81.
22. Rustgi AK, Peppercorn MA. Gluten sensitive enteropathy and systemic lupus erythematodes.
Arch Intern Med 1988; 148: 1583-6.
23. Mulder CJJ, Tytgat GN, Groenland
F, Pena AS.
Combined Celiac disease and thyroid disease, a study od
17 cases. J Clin Nutr Gastroeterol 1988; 3: 89-94.
24. Collin P, Salmi J, Hallstrom O et
al. High frequency of celiac disease in adult patients with typte I diabetes. Scand J Gastroenterol 1989; 24: 89-93.
25. Katz SI. Hall
RP, Lawley TJ, Strober W. Dermatitis herpetiformis of the skin and the gut.
Ann Intern Med 1980; 92: 857-61.
26. Kristoferisch W, Pointner H.
Progressive cerebelar syndroma in adult celiac disease. J
Neurol 1987; 234: 116-19.
27. Cooper BT, Holmes GK, Cooke WT.
Lymphoma risk in coelac disease of later life. Digestion
1982; 23: 89-92.
28. Mathus-Vliegen EMH, Van Halteren
H and Tytgat GNJ. Malignant lymphoma in celiac disease: Various manifestation with distinct symptomatology and prognosis. J
Intern Med 1994; 236:43-8..
Značaj odredjivanja alveolo-arterijskog gradijenta kod
bolesnika sa cirozom jetre
SAŽETAK
Uočeno je da
alveolo-arterijski gradijent ≥ 2 kPa sa ili bez
hipoksemije ukazuje na intrapulmonarnu vaskularnu dilataciju. Za procenu značaja
alveolo-arterijskog gradijenta kod pacijenata sa
cirozom jetre analizirali smo 70 pacijenata. Pritisak kiseonika u alveolarnom
vazduhu izračunat je na osnovu izmerenog
atmosferskog pritiska i ugljen-dioksida u izdahnutom vazduhu, dok je vrednost
Pa, 02 bila izmerena direktno u arterijskoj krvi. Ortodeoksija je potvrđena
kod 10 (14,3%) hipoksemičnih i 4 (5,7%)
normoksemiča pacijenta. U našoj studiji 30 (42.8%)
pacijenata imali su alveolo-arterijski gradijent preko 2 kPa. Svi
pacijenti sa ortodeoksijom imali su povišene vrednosti
alveolo-arterijskog gradijenta, čija je srednja vrednost u ležećem
položaju bila 4.03 ± 2.36 kPa, dok je u sedećem ta vrednost iznosila 5.73
± 2.65 kPa. Kod pacijenata sa restriktivnim ventilatornim poremećajima
izazvanim ascitesom, koji nisu imali ortodeoksiju, srednja vrednost P(A-a) 02 u ležećem položaju iznosila je 2.77 ± 1.12 kPa, dok je u
sedećem iznosila 1.80 ± 1.04 kPa. Rezultati ukazuju da
su ortodeoksija i povećan alveolo-arterijski gradijent senzitivni
funkcionalni parametri u diagnostici intrapulmonalne vaskularne dilatacije koja
predstavlja glavni patogenetski mehanizam u nastanku teških respiratornih
poremećaja u cirozi jetre.
UVOD
Kod pacijenata sa cirozom jetre i izraženom hipoksemijom, IPVD i desno-levi
šantovi, predstavljaju glavni patogenetski mehanizam u nastanku teških
respiratornih poremećaja (1-3).
Najčešći
plućni simptom jeste dispnea koja se javlja kod skoro 40% pacijenata sa IPVD. Preko 70% pacijenata predviđenih za
transplantaciju jetre žali se na dispneu (4). Druga
tipična manifestacija IPVD u cirozi je platipnea, koja označava
dispneu izraženiju u stojećem ili sedećem u
odnosu na ležeći položaj (5). Ortodeoksija je još jedan klinički
pokazatelj IPVD, koja predstavlja pad parcijalnog pritiska kiseonika za više od 10%, kad pacijent pređe iz ležećeg u sedeći
položaj.
Hipoksemija sa
parcijalnim pritiskom kiseonika (Pa,02) manjim od 8
kPa u odsustvu drugih kardio-respiratornih bolesti ukazuje na postojanje IPVD
(6,7).
Određivanje P(A-a)02 najjednostavniji
je metod za procenjivanja ukupnog odnosa ventilacije i perfuzije u plućima.
Uočeno je da P(A-a)02 ≥ 2 kPa sa ili bez hipoksemije ukazuje na postojanje
IPVD (8).
PACIJENTI
I METODE
U prospektivnoj
studiji analizirali smo 70 pacijenata sa cirozom
jetre, koji su lečeni na Institutu za digestivne bolesti i Institutu za
plućne bolesti Kliničkog centra Srbije. Gasne analize rađene su
aparatom Blood Gas Manager IL 1312, dok su predviđene vrednosti za
parcijalni pritisak kiseonika u krvi izračunate pomoću Sorbinijeve
jednačine (Pa,02 =103,5
-0,42 x godine) (9). Anallizu gasova u arterijskoj krvi vršili
smo u ležećem i sedećem položaju, pri udisanju sobnog vazduhu i nakon
15 minuta udisanja hiperoksične smese, direktnom metodom, koja podrazumeva
uzimanje aretrijskog uzorka krvi hepariniziranim špricem.
Spirometrijska
merenja u našoj studiji su bila izvedena pomoću otvorenog spirometrijskog
sistema sa pneumotahografijom (Pneumoscreen II
spirometar) za određivanja: vitalnog kapaciteta (VK), forsiranog vitalnog
kapaciteta (FVK) i forsiranog ekspirijumski volumen u prvoj sekundi (FEVı).
Telesnom
pletizmografija (Bodyscreen II) merili smo: totalni plućni kapacitet
(TPK), toraksni gasni volumen (TGV), rezidualni volumen (RV) i otpor strujanju
vazduha u disajnim putevima putevima (RaW).
Alveolo-arterijski
gredijent pritiska kiseonika (P(A-a)02 = PA02 – Pa,02)
izračunavali smo na način koji se uobičajeno koristi u praksi.
Pritisak kiseonika u alveolskom vazduhu izračunat je na osnovu izmerenog atmosferskog
pritiska i pritiska ugljen-dioksida u izdahnutom vazduhu (PA02 = Fi02 x (Pb –PaH2O) – PA,co2 (Fio2 + 1 – FiO2/ Ra) (FiO2 – frakciona inspiratorna
koncentracija kiseonika; Ra – respiratorni količnik), dok je Pa,02 meren
direktno u arterijskoj krvi (10).
S obzirom na
promenjivost respiracijskih gasova arterijske krvi kod obolelih od ciroze jetre
pri promeni položaja tela, promene P(A-a)02 pri prelasku iz ležećeg u sedeći položaj, za
najmanje 0.66 kPa, predstavljaju signifikantan znak IPVD (11).
Plućnim
funkcionalnim testovima određivali smo transfer faktora (TL,CO) i koeficijent transfera (KCO = TL,CO podeljen sa efektivnim alveolarnim
volumenom), kao pokazatelje stanja alveolo-kapilarne difuzije Parametre
difuzije merili smo ugljen-monoksidom metodom jednog udaha, sa Transferscreen
aparatom. Donju granicu normale za transfer faktora dterminisali smo matematičkim
putem tako što smo od predviđenih vrednosti za TL,CO oduzeli 1.64 SD (SD za muškarce 1.42, SD za
žene 1.17). Predviđene vrednosti TL,CO računali smo na osnovu usvojenih standarda (za muškarace
predviđeni TL,CO = 11.11 x visina u metrima - 0.066 x godine
– 6.03; za žene, predviđeni TL,CO =
8.18 x visina u metrima – 0.049 x godine – 2.74) (11).
Restriktivne
ventilatorne poremećaje određivani su na
osnovu spirometrijskih parametara: VK, indeksa 100 x FEVı / VK (Tiffeneau)
i UPK. Donja granica normale determinisana je kao predviđena vrednost –
1.64 SD (za muškarace SD za FVK = 0.61, SD za UPK =0.70, i za Tiffeneau
SD =7.17; za žene SD za FVK = 0.43, SD za UPK
= 0.60, i za Tiffeneau SD = 6.51) (12).
Statističke
analize izvedene su korišćenjem x2 i t-testa.
REZULTATI
Ortodeoksija
je potvrđena kod 10 hipoksemičnih (14.3%) i kod 4 (5.7%) normoksemična
pacijenata. U uslovima
udisanja sobnog vazduhom srednja vrednost Pa,02 u
ležećem položaju iznosila je 9.40 ± 1.74 kPa, dok je u sedećem
položaju prosečna vrednost Pa02 bila
7.41 ± 1.81 kPa. U uslovima udisanja 100% kiseonika srednja vrednost Pa02 u ležećem položaju iznosila je 35.49 ± 17.95 kPa, dok je
u sedećem položaju prosečna vrednost Pa02 bila
21.07 ± 14.41 kPa (Tabela 1).
U našoj studiji
30 (42.8%) pacijenata su imali P(A-a)02 preko 2 kPa. Svi pacijenti sa ortodeoksijom imali su povećan
P(A-a)02, čija je prosečna vrednosti u
ležećem položaju iznosila 4.03 ± 2.36 kPa, dok je u sedećem položaju
bila 5.73 ± 2.65 kPa. Pacijenti sa ortodeoksijom i povećanim P(A-a)02 imali
su IPVD. Pacijenti sa ortodeoksijom i normoksemijom takođe su imali povećanu
vrednosti P(A-a)02, koja se kretala od 2.1 do 2.37 kPa što prezentuje subkliničku
IPVD.
Dijagnoza
ascitesa potvrđena je kod 43 (61%) pacijenata. Restriktivni ventilatorni poremećaji
i povećan P(A-a)02 dijagnostikovani su kod 16 (37.2%) pacijenata sa ascitesom.
Srednja vrednost parametara restrikcije iznosila je: VK 73.0 ± 14.4, UPK 83.7 ±
13.5, a Tiffeneau indeks 78.3 ± 3.37.
Kod pacijenta sa
restriktivnim ventilatornim poremećajem, bez ortodeoksije, srednja
vrednost P(A-a)02 u ležećem položaju iznosila je 2.77 ± 1.12 kPa, a u sedećem
1.80 ±1.04 kPa. Upoređujući pacijente sa i bez ortodeoksije,
uspostvaljena je statistički značajna razlika u vrednostima P(A-a)02 (p
= 0.001, t-test), posebno u sedećem položaju (Tabela 2).
Snižen transfer
faktor (TL,CO)
registrovan je kod 38 (54%) pacijenata, dok je snižen koeficijent transfera (KCO) zabeležen kod 46 (66%) pacijenata. Srednja vrednost
transfer faktora bila je 6.24 (65.72%). Kod svih pacijenata sa
IPVD i subkliničkom IPVD manifestovale su se snižene vrednosti transfer
faktora i koeficijenta transfera.
Povećan P(A-a)02 i normalan transfer faktor imalo je 11 (16%)
(pacijenati sa restriktivnim disajnim poremećajima), dok je povećan P(A-a)02 i
snižen transfer faktor imalo 14 (20%)) pacijenata (pacijenti sa IPVD). Snižen
transfer faktor i normalan P(A-a)02 registrovan je kod 17 (24%) pacijenata. Nisu pronađene
statistički signifikantne razlike između povećanih vrednosti P(A-a)02 i
poremećaja difuzije (x2 - test, p =.62) (Tabela3).
DISKUSIJA
Bashour
i Cochran, 1966.
godine, ispitujući 26 pacijenata sa cirozom
jetre, izmerili su srednju vrednost alveolo-arterijskog gradijenta 5.97 kPa
(13).
U grupi
pacijenata predviđenih za proto-kavalni šant, Naeije i saradnici (1985)
nalaze srednju vrednost P(A-a)02 od 4.85 kPa (14).
Hourani
i saradnici, 1991.
godine, saopštavaju povećan gradijent pritiska
kiseonika u 45% pacijenata predviđenih za transplantaciju jetre, sa
srednjom vrednosti P(A-a)02
4.89 kPa (4).
Ističući
značaj ovog funkcionog poremećaja, Fahy i saradnici (1992), navode da
je 69% pacijenata, predviđenih za transplantaciju jetre, imalo povećan
P(A-a)02 (15).
Behera
i saradnici, 1998.
godine, nalaze hipoksemiju s povećanjem P(A-a)02 u 26.7% pacijenata sa cirozom jetre. Bolje rezultate imali su
pacijenati sa ekstrahepatičnom opstrukcijom
portne vene (16).
U Falonovoj
studiji od 207 kandidata predviđenih za transplantaciju jetre 66% su imali
vrednost P(A-a)02 veću od 2 kPa (17).
U našoj studiji
30 (42.8%) pacijenata je imalo P(A-a)02 preko 2 kPa. Povećana vrednost P(A-a)02 (5.73 kPa ± 2.65), posebno u sedećem položaju, bila je
zabeležena kod svih pacijenata sa ortodeoksijom, što ukazuje na IPVD.
Često
hiperventilisanje pacijenata s cirozom jetre može da poveća vrednosti P(A-a)02 čak
i u slučaju normoksemije. Zbog toga je, P(A-a)02 senzitivan parametar poremećaja gasne razmene u plućima.
Veliki dijagnostički značaj ovih testova potvrđuje i nalaz
neizmenjenog bazičnog parcijalnog pritiska kiseonika pri postojanju
subkliničke IPVD nezavisno od povećanja P(A-a)02 (18).
Subklinička
IPVD, kao poseban entitet, predstavlja tranzitornu fazu u razvoju
hepatopulmonalnog sindroma (HPS). Uprkos IPVD i porastu P(A-a)02 parcijalni pritisak kiseonika u arterijskoj krvi može dugo
vremena da ostane nepromenjen. Ovo je rezultat alveolne hiperventilacije, hiperdinamske
cirkulacije i porasta minutnog volumena srca što je karakteristično za
cirozu (19,20).
Subklinička
vazodilatacija u plućima, praćena normoksemijom, može biti otkrivena
u ranoj fazi ciroze jetre. Sa progresijom plućnih promena
hipoksemija se sve više ispoljava. Tokom bolesti,
oksigenacija može da se pogoršava i bez pogoršavanja funkcije jetre (21).
U našoj studiji,
sva četiri pacijenta sa subkliničkom IPVD bili su normoksemični
nezavisno od povećane vrednosti P(A-a)02 koja se kretala od 2.1 do 2.37 kPa.
Kod pacijenata sa
ventilatornim restriktivnim poremećajima izazvanim ascitesom, koji nisu
imali ortodeoksiju, srednja vrednost P(A-a)02 u ležećem položaju bila je 2.77 ± 1.12 kPa dok je u sedećem
iznosila 1.80 ±1.04 kPa.
Pacijenti sa izraženim ascitesom u stojećem položaju obično
imaju normalne vrednosti respiratornih gasova u arterijskoj krvi, dok se
parcijalna insuficijencija respiracije manifestuje u ležećem položaju. Prerano zatvaranje disajnih puteva uslovljava da veliki delovi
donjih plućnih regiona ne učestvuju u ventilisanju, dok u isto vreme
imaju očuvanu perfuziju, tako da se bitno pogoršava
ventilaciono-perfuzioni odnos. Perfuzija u
neventilisanim delovima, uslovljava da znatna količina krvi prolazi kroz
pluća a da se ne oksigeniše (22).
U studiji, Changa
i saradnika (1997), ispitivan je efekat ascitesa na
plućnu funkciju kod dve grupe pacijenata, upoređivanjem terapijskih
efekata paracenteze i diuretika. Za razliku od pacijenata lečenih
paracentezom, pacijenti koji su lečeni diuretskom terapijom pokazali su
znatno poboljšanje gasne razmene uz povećanje Pa,02
i veliko smanjenje P(A-a)02. Poboljšanje
oksidenacije upotrebom diuretika pokazuje da osim mehaničkog efekta
ascitesa intersticijalni plućni edem i retencija tečnosti dodatno
doprinose pogoršavanju gasne razmene (23).
Ispitivanja
zasnovana na udisanju 100% kiseonika takođe često
se koriste kod pacijenata sa dokazanom ili suspektnom IPVD. Na osnovu
oksigenacionog odgovora nakon udisanja 100% kiseonika, opisana su dva tipa
vaskularnih abnormalnosti kod pacijenata sa
hepatocelularnom insuficijencijom. Tip-I karakteriše difuzna vaskularna
dilatacija na prekapilarnom nivou, dok je Tip-2
definisan pravim anatomskim plućnim arterio-venskim šantovima. Primena
100% kiseonika dovodi do značajnog povećanja parcijalnog pritiska
kiseonika kod Tipa-I, dok je efekat minimalan kod Tipa-2 (7).
Nakon udisanja
100% kiseonika, kod nekih pacijenata, postiže se nepotpuni oksigenacioni
odgovor sa parcijalnom korekcijom hipoksemije. Poremećaj
je shvaćen kao posledica IPVD, što takođe doprinosi povećanju
šantne frakcije, a označava se terminom difuziono-perfuzioni defekt ili alveolo-kapilarni disekvilibrijum kiseonika (24, 25).
Hourani
i saradnici, 1991.
godine, opisuju da je od 116 pacijenata sa cirozom
jetre 60 (52%) imalo smanjenje difuzionog kapaciteta. Povećan kapilarni
plazmatski volumen i alveolna kapilarna dilatacija dovode do porasta difuzione
distance, što predstavlja osnovni mehanizam poremećaja transfera
ugljen-monoksida i kiseonika u oboljenjima jetre. Smanjenje
difuzionog kapaciteta kod normalno oksigenisanih pacijenata takođe se može
objasniti postojanjem IPVD, odnosno subkliničke IPVD. Značajno
je da kod 40% pacijenata iz ove studije, koji su imali poremećaje
difuzije, nije pronađen porast P(A-a)02 (4).
Krowka
i Cortese, 1994.
godine, navode da u nastanku izolovanog smanjenja TL,CO pored IPVD, učestvuju i drugi patogenetski mehanizmi,
kao: difuzne intersticijumske plućne bolesti koje u početnoj fazi ne
izazivaju restriktivne poremećaje, protok krvi kroz neventilisane alveole,
ventilaciono-perfuzioni disbalans i/ili druge plućne vaskularne bolesti
(7).
U našoj studiji,
snižen transfer faktor i normalan P(A-a)02registrovan je kod 17 (27%) pacijenata. Povećan P(A-a)02 i
normalan transfer faktor imalo je 11(16%) pacijenata, dok je povećane P(A-a)02 i snižen transfer faktor imalo 14 (20%) pacijenata (slučajevi
sa IPVD). Nije bilo signifikantne razlike između ove dve grupe, što
ukazuje da izolovano smanjenje TL,CO ne može da se objasni samo postojanjem IPVD.
Ortodeoksija i
povećan P(A-a)02 su senzitivni funkcionalni parametri u diagnostici IPVD, koja
predstavlja glavni patogentski mehanizam u nastanku teških respiratornih poremećaja
u ciroze jetre.


