|
|
Alimentary
tract and pancreas Alimentarni
trakt i pankreas |
||
|
1Thanaa El-Sayed Helal, 2Nehal Mohamed El-Koufy, 2Mohamed Khalil Abdel Khalek, 3Manal Hassan Moussa, 4Hesham Abdel Kader Hassan. 1Department of Pathology, Faculty of Medicine, Ain Shams University, Cairo, 2Department of Padiatrics, Faculty of Medicine, Cairo University, Cairo, 3Department of Histology, Ain Shams University, Cairo, 4Departent of Paediatric Gastroenterology and Hepatology, State University of New York, Health Science Center, Syracuse , NY,USA. . |
ARCH
GASTROENTEROHEPATOL 2003; 22 (No 1 - 2): 12 – 17 Morphometric
and DNA analysis
in Egyptian children with
chronic liver diseases Morfometrijska
i DNA analiza Egipatske dece sa hroni~nim
bolestima jetre (
accepted April 24th, 2003 ) |
||
|
Key Words: chronic liver disease, morphometry, DNA analysis, dysplasia. |
Abstract The
aim of the present study was to assess the value of morphometric and DNA
image analysis in the assessment
of chronic liver diseases in children. Seventy liver biopsies from children
with chronic liver diseases
were included in the study (30 infantile cholestasis, 24 chronic hepatitis,
16 glycogen storage disease,
GSD). The biopsies were examined histologically for estimation of the grade
of the disease activity,
stage of fibrosis, and degree of dysplasia. DNA ploidy was determined by
using Feulgen,s stained
sections and CAS 200 image analyser. Morphometric analysis of the nuclear
area, nucleo-cytoplasmatic (N/C)
ratio and the degree of fibrosis (fibrotic index) were also estimated.
Although the grade of
disease activity was not significantly diferent among the 3 disease group,
the stage of fibrosis was significantlly more
in the cholestatic and hepatitis group than GSD. DNA aneuploidy was observed
in 8 out
of 70 liver biopsies, 4 with infantile cholestasis, and 4 with chronic
hepatitis. The 3 disease groups were
not statistically different regarding the nuclear area or the fibrotic index.
However, the N/C ratio was
significantly higher in the cholestatic and hepatitis group than in GSD.
Moreover, the semiquantitative staging
of fibrosis was significantly correlated with the qantitative measurement of
the fibrotic index.
These results suggest that image analysis especially morphometry is a helpful
objective method in
the investigation of chronic liver diseases in children. Liver cell
dysplasia, DNA aneupoloidya, and icreased
N/C ratio can be used as risk factor for developing hepatocellular carcinoma
in children with chronic
liver diseases either early or later in the life. Infantile cholestasis seems
to have premalignant potential
in Egyptian children similar to well known role of chronic viral hepatitis. Sa`etak Cilj ove
studije je bio da se proceni vrednost morfometrijske i DNA analize u proceni
hroni~nih bolesti jetre u
dece. Sedamdeset biopsija jetre dece sa hroni~nim bolestima jetre je
uklju~eno u ovo ispitivanje (30 slu~ajeva
infantilne holestaze, 24 hroni~nog hepatitisa, l6 glikogenoza, GSD). Biopsije
su pregledane histolo{ ki i
izvr{ena je procena stepena aktivnosti bolesti, stadijum fibroze, i stepen
displazije. DNA-ploidija je
odredjivana kori{}enjem Feulgen-ovim rastvorom bojenih sekcija tkiva jetre i
CAS 200 analizatorom slike.
Morfometrijska analiza jedra, nukleo-citoplazmatskog (N/C) odnosa i stupnja
fibroze (indeks fibroze)
je takodje vr~ena. Premda stadijum aktivnosti bolesti nije bio zna~ajno
razli~it izmedju ispitivane tri
grupe bolesti, stupanj fibroze (fibroti~ki indeks) je bio izra`eniji u grupi
holestaznih bolesti jetre i hepatitisima nego u
GSD. DNA aneuploidija je otkrivena u 8 od 70 biopsija, i to u 4 sa
infantilnom holestazom i 4 sa
hroni~nim hepatitisom. U sve tri grupe bolesti jetre nije bilo razlika u
pogledu procene jedra i fibroti~kog
indeksa. Medjutim N/C odnos je bio zna~ajno ve}i u dece biopsijskim uzorcima
dece sa holestazom
i hepatitisom mego u onih sa GSD. Semikvantitativno merenje stadijuma
odmaklosti firboze je bilo u
signifikatnoj korelaciji sa kvantitaivnim merenjem fibroti~kog indeksa. Ovi
rezultati ukazuju da posebno
morfometrijska analiza uzoraka tkiva jetre je dobra pomo} u ispitivanju dece
sa hroni~nim bolestima jetre.
]elijska displazija, DNA aneuplodija, i povi{en N/C odnos je faktori rizika
za nastanak hepatocvelularnog karcinoma
jetre u dece sa hroni~nim bolstima jetre. Infantilna holestaza je ~ini se
grupa bolesti
sa najvi{im premalignim popterncijalom u Egipatske dece sli~no virusnim
hepatitisima. |
||
|
Klju~ne
re~i: hroni~ne
bolesti jetre, morfometrija, DNA analiza,
displazija. |
|||
|
|
Several
studies have investigated the preneoplastic changes
in hepatocytes in chronic liver diseases in the adults.
Morphologic studies indicate that dysplasia is frequently associated
with the development of hepatocelullar carcinoma
(HCC) (1-4). Measurement of DNA content in patients
with chronic hepatitis have revealed a significant correlation
betwen the DNA index and the histological evidence of
HCC (5,6). Morphometric analysis of liver cells have
shown that increased nuclear area and/or nuclearcytoplasmatic ratio
represent the more likely candidate for precancerous
changes in the life (7,8). Although
these preneoplastic conditions have been extensivelly
studied in chronic liver diseases in adults, similar
studies in the paediatric age group are rare (1-8). Available
data consist of children with chronic HCV (Hepatitis
C virus) infection and they were primarily clinically and
morphologically oriented with no emphesis on the
premalignant changes (9-11). Esquivel
reviwed the histopathology of liver from 72 children
with various causes of chronic liver diseases. He detected
9 cases (12.5%) with HCC and/or liver cell dysplasia (12).
The primary liver diseases in these 9 children was
tyrosinemia (3), biliary atresia (2), chronic virus hepatitis
(2), neonatal hepatitis (1), and neonatal iron storage disease
(1). This sudy indicated that children with chronic
liver diseases may develop HCC early in the life or show
dysplastic changes which may proceed to carcinoma later
in the life. The
objective of the current study is to compare the possible
preneoplastic changes including dysplasia, nuclear
mophometric features and DNA abnormalities in various
form of chronic liver diseases in Egyptian children. MATERIAL
AND METHODS Patients
Seventy children diagnosed as having chronic liver
disease at the Paediatric Hospital, Cairo University, Egypt
were selected to constitute the subject of the current study.
Those children underwent careful clinical, radiological, laboratory
investigations and liver biopsy. Consequently, they
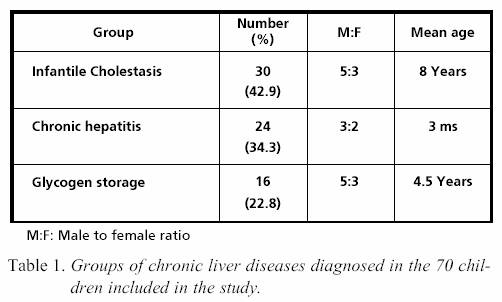
were classified into 3 groups. Table 1. The first group was
infantile cholestasis. It consisted of 30 children who were diagnosed
as neonatal hepatitis (19), paucity of bile ducts (8 cases),
and extrahepatic biliary atresia (3 cases). The second group
included 24 patients with chronic hepatitis. Viral hepatitis
was confirmed in 9 of these patients (8 were hepatitis C,
1 hepatitis B). Autoimmune hepatitis was diagnosed in 8;
cryptogenic hepatitis in 7 patients. The third group consisted of
16 children with glycogen storage disease. Histopathological
study True cut needle liver biopsies were
obtained from all children which enrolled this study.
They
were fixed in buffered formalin and embedded in paraffin.
Tissue sections were stained with haematoxylin and
eosin (H&E), periodic acid Schill (PAS), and Massonís trichrome
stains. These sections were examined to confirm the
diagnosis, estimate the grade of disease activity, and stage
of fibrosis according to Scheuer (13). Moreover, the presence
of dysplasia was assessed as recommended by the International
Working Party (14). DNA
image anaysis Tissue sections from paraffinembedded liver
tissue were cut on poly-L-lysine coated slides.
These tissue sections were stained by Feulgen technique using
the quantitative DNA staining kit (Becton Dickinson,
Chicago,USA). Feulgen-stained slides were analysed
on the CAS200 image analyser (Becton Dickinson, Chicago,USA),
using quantitative DNA analysis software programme.
For each specimen, at least 200 non-overlapping nuclei
were analysed. Whenever possible, about 20 lymphocytes
were also calculated to be used as an internal diploid
control. The DNA content was computed as the integrated optical
density of the Feulgen-stained nuclei (15). DNA
index (DI) of the studied tumours with the mean peak was
in the DI range of 0.9-1.1 (2C peak) or aneuploid tumours
with the DI fell outside the diploid range or more than
20% of the cells were present in the G2/M phase (16). DI
was computed by dividing the model mass value of the test
cells by the known DNA content value of the control cells
in picograms (7.18pg) (17). Morphometric
analysis The nuclear features of liver cells
including nuclear area and nucleo-cytoplasmatic ratio (N/C)
were estimated by using H&E stained sections and computerised
image analyser (Cell Analysis System, Becton Dickinson,Chicago,USA).
For each case, the cell measurement program
ws applied to count randomly the nuclear features of
300 hepatocytes at magnification x400. Then, the mean
nuclear area and mean N/C ratio were calculated. Quantitative
measurement of the severity of liver fibrosis was
achieved by using the same computerised image analyser
and Masonís trichrome-stained sections. The
fibrotic index
(FI) was calculated for each case as the ratio of the area
of fibrosis to the area of the entire tissue specimen (18). Statistical
analysis Unpaired T-test was used to compare the
3 categories of chronic liver diseases regarding disease
activity, stage of fibrosis, morphometric nuclear features
and fibrotic index. The 3 disease categories were also
compared with respect to dysplasia and DNA ploidy by
applying Chi-square test. Correlation between the grade of
disease activity and stage of fibrosis as well as between the
semiquantitative score of liver fibrosis (stage of fibrosis) and
the quantitative score of fibrosis (fibrotic index) was
assessed by Pearson,s correlation coefficient. RESULTS Histopathological
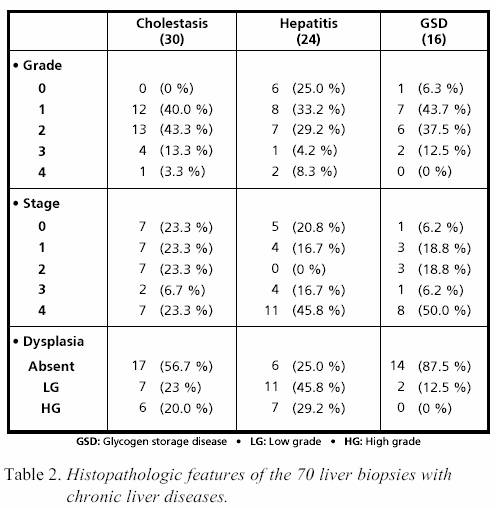
data The group of infantile cholestasis consisted
of 30 children. Their liver biopsies were diagnosed as
neonatal hepatitis (19), paucity of bile ducts (8), and
extrahepatic biliary atresia (3). Evidence of cirrhosis (stage
4) was found in 7 liver biopsies (23.3%). The group of
chronic hepatitis (CH) included 24 children. The aetiology of
hepatitis was viral in 9 cases (8 hepatitis C virus, 1 hepatitis
B virus ), autoimmune (8), and cryptogenic (7). Eleven
out of 24 liver biopsies (45.8%) showed cirrhosis. The
last group in this study was glycogen storage diseases (GSD)
which consisted of 16 children. Eight of their liver biopsies
(50%) showed cirrhosis (Table 2). The
grade of disease activity was not significantly different among
the 3 disease groups. The stage of fibrosis was
significantly positive higher in the cholestatic group than
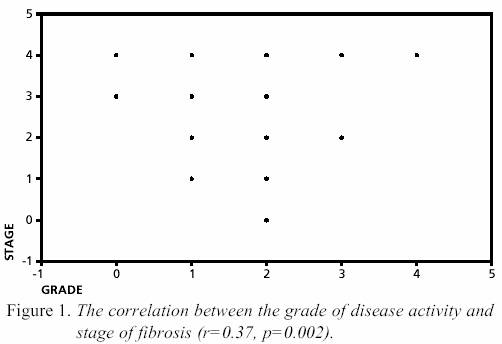
in GSD (T=2, p=0.05). A highly significant positive correlation
was obtained between the grade and stage
(r=0.37,p=0.002).
Figure 1. The frequency of dysplasia was
significantly more in the cholestatic group (x2=10.8, p<0.01)
and chronic hepatitis (x2=14.5, p<0.01) than in GSD.
However, no significant difference was detected between
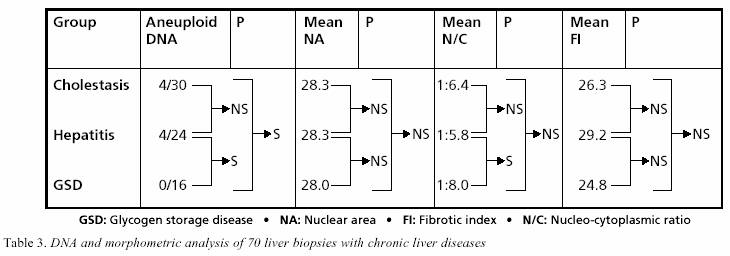
the former two groups. DNA
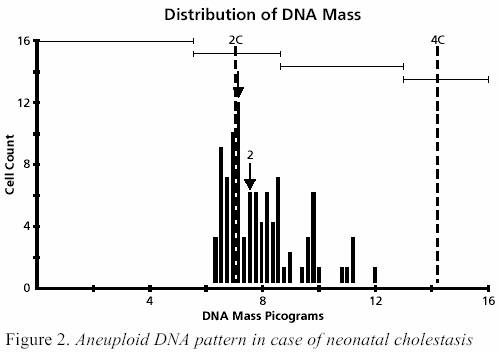
image analysis (Table 3) Aneuploid cell populations were
seen in 8 out of 70 paediatric cases with chronic liver
diseases (11.4%) with DNA index (DI) ranging between
1.18 and 1.51 (mean 1.25). All these 8 biopsies had
the morphologic features of dysplasia Four
out of 8 aneuploid biopsies were the cholestatic group
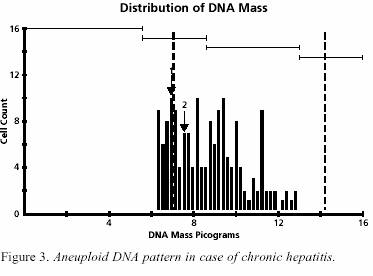
(Figure 2) and the other 4 were of the hepatitis group (Figure
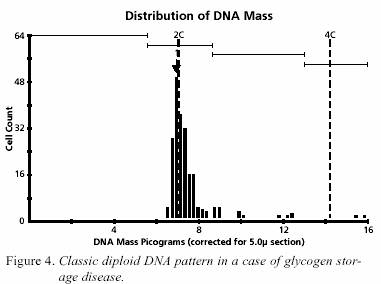
3). All 16 liver specimens with GSD displayed the classic
diploid pattern (Figure 4) with minor variation for the
DI. Morphometric
anaysis The group of infantile cholestasis, chronic
hepatitis and GSD were not statistically different regarding
the nuclear area and the fibrotic index. However,
N/C ratio was significantly higher in the choelstatic group
(T=2.4, p=0.02) and in the hepatitis group (T=3.3,
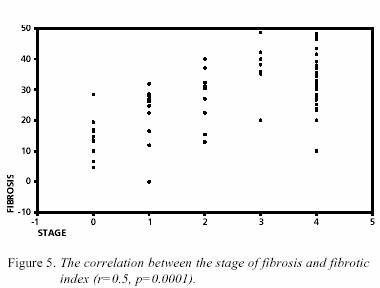
p=0.003) than in the GSD group. There was a highly
significant correlation between the semiquantitative (staging)
and the quantitative (FI) estimation of fibrosis (r=0.5,
p=0.0001). Figure 5.
DISCUSSION The
commonest aetiology of chronic liver diseases in this
study of 70 Egyptian children with chronic liver diseases is
infantile cholestatic syndromes (43%). Conversely, viral
hepatitis accounted only 13% of the total cases. This findings
is consistent with North American, European, and Indian
reports which suggest that the incidence of chronic hepatitis
B and C is
of lower frequency in childhood (20,21).
Our results are in parallel with other Egyptian population
based studies which indicate that chronic HCV is
much rarer below the age of 20 years (8%) (22). Assessment
of the grade of disease activity and stage of fibrosis
in the liver biopsies from children with viral hepatitis
revealed that although they demonstrate low grades
of disease activity, the stages of fibrosis are much higher.
This agrees with Badizadegan who suggested that in
spite of mild histological necroinflammatory activity, the
stage of fibrosis in children with HCV hepatitis
can be more
pronounced (9). In
the study of the international childhood cancer incidence of
coordinated by the Agency for Research on Cancer,
hepatocelullar carcinoma (HCC) in children was most
common in parts of Asia and Africa including Egypt (19). The
histological, morphometric and flow-cytometric investigations
are increasingly defining the preneoplastic hepatic
lesions and the stepwise evolution of HCC in adults.
However, the search for similar studies in children are
lacking. This might be related to presumed small incidence of
HCC in childhood and small number of well documented cases
(23,23,25). However, in the last few years the
incidence of HCC in children seems to be rising. This necessitates
selection of children with chronic liver diseases who
are prone to develop hepatic malignancies and further
on deserve careful follow up. We
find that the frequency of hepatocelullar dysplasia is
significantly higher in children with infantile cholestasis and chronic
hepatitis than in those with GSD. Since liver cell
dysplasia has been claimed to play a role in the evolution of
HCC, our results suggest that infantile cholestasis and
chronic hepatitis have a more tendency for malignant transformation
than GSD (1-4). This is supported by the observation
that dysplastic liver cells show aneuploid DNA
pattern as revealed in the current study and others (2,5,6,29).
It is worth to mention that the importance of viral
hepatitis as potential risk factor for malignancy has been
previously reported in several studies ( 8,30-33). Similarly,
the absence of dysplasia and aneuploidy in cases of
GSD is supported by the tendency of GSD to progress to
hepatocelullar adenoma rather than carcinoma (34). Nevertheless,
our observation that infantile cholestasis has similar
risk for developing malignancy as chronic hepatitis has
not been reported previously. This is to support comparable increase
in the N/C ratio in the cholestatic and hepatitis
groups which were significantly higher than N/C ratio
in the GSD group. The reported cases of HCC in children with
infantile cholestasis are only confined to cases with
familial intrehepatic cholestasis syndromes which constitute
about one fourth of the cases in the colestatic group
in this study (35-37). The majority of the children in
this
group had neonatal hepatitis (NH) which was not an antecedent
of HCC in childhood. However, Moore recently reported
a child who was diagnosed as having NH at 4th months
of life, developed cirrhosis at 23rd months of age, and
HCC at 28th month (38). Thus, it is belived that NH may
be one of paediatric chronic liver disease tham may eventually
progress to HCC. Therefore, follow up of children suffering
of NH may be recommended especially in those
developing cirrhosis and/or dysplasia with aneuploid DNA
pattern. In fact, 4 out of 19 children included in this study
showed cirrhosis and another 2 children revealed high
grade of dysplasia with aneuploid cell populations in their
liver biopsies. One
of the important points in the current study is the morphometric
estimation of the fibrotic index. This quantitative measurement
of fibrosis was well correlated with the
semiquantitative measurement of the stage of fibrosis. This
finding whish is supported by several reports indicates that
image analysis is simple, rapid and reproducible tool
for the objective quantification of liver fibrosis (39- 41).
Although the assessment of liver dysplasia, DNA ploidy
and N/C ratio revealed that these potential premalignant changes
were more in the cholestatic and chronic hepatitis
children than those with GSD, the fibrotic index was
not significantly different among our 3 groups. This result
may indicate that the degree of firbrosis in not necessarly a
measure of the preneoplastic potential of the chronic
liver disease. In the other words, cirrhosis might be an
essential event in the multistep evelution of HCC in children
as well. This is supported by stidy of Berman who founded
that cirrhosis is found in only 15% of HCC in children (42).
Yet, large studies are necessary to further define this
proposal. Finally,
the current preliminary study imply that chronic viral
hepatitis appears to constitute a minor percentage of
chronic liver diseases in Egyptian children. In order to predict
the prognosis and objectively measure liver fibrosis degree,
image analysis especially morphometry is an important
tool. Our stady further indicates that infantile cholestasismay
have some premalignant potential similar to
the chronic viral hepatitis. We found that liver cell dysplasia, DNA
aneupolidy and increased N/C ratio can be used
as risk factor for developing HCC in children with chronic
liver diseases. Conversely, the value of the fibrotic index
as indicator of high risk paediatric cases with liver diseaes
was not confirmed in this study. |
||
|
|
REFERENCES: 1.
Anthony
PP, Vogel CL, Barker LF. Liver cell dysplasia: A premalignant condition. J Clin Pathol 1973;26:217-23. 2.
Thomas
RM, Berman JJ, Yetter RA et al. Liver cell dysplasia: A DNA aneuploid lesion with
distinct morphologic features. Hum Pathol
1992;3:496- 503. 3.
Borzio
M, Bruno S, Roncalli M et al. Liver cell dysplasia is a major risk factor for hepatocellular carcinoma in cirrhosis. A prospective
study. Gastroenterology 1995; 108:812-7. 4.
Karim
B, Alex G, Smith AL, Hardikar W. Hepatitis C infection in children: A Melbourne
perpective. J Paediatr Child Health 2000;36:385-8. 5.
Hoshiyama
A, Kimura A, Fujisawa T et al. Clinical and histologic features of
chronic hepatitis C virus after blood transfusion in
Japanese children. Pediatrics 2000;105;62-5. 6.
Esquivel
C, Gutierez C, Cox KL et al. Hepatocellular carcinoma and liver cell
dysplasia in children with chronic liver
diseases. J Pediatr Surg 1994;29:1465-9. 7.
Scheuer
PJ. Classification of chronic viral
hepatitis: A need for reassessment. J Hepatol 1991;13:372-4. 8.
International
Working Party. Terminology of nodular hepatocellular lesions.
Hepatology 1996;22:983-93. 9.
Bacus
JW, Grace LJ. Optical microscope system for standardised cell measurements and
analyses. Appl Optics 1987;26:3280-93. 10.
El
Sheikh TM, Silverman JF, McCool JW, Riley RS. Comparative DNA analysis of solid tumours by flow-cytometric and image analysis
of touch imprints and flow suspension. Am J Clin
Pathol 1992;98:296-304. 11.
Taylor
SR, Ernstoff T, Stitely S. Central
values and variations of measured nuclear DNA
content in imprints of normal tissues determined
by image analysis. Cytometry 1989;10:382-7. 12.
Kage
M, Shimamatu K, Nakashima E et al. Long term evolution of fibrosis from chronic
hepatitis to cirrhosis in patients with hepatitis
C: Morphometric analysis of repeated
biopsies. Hepatology 1997;25:1028-31. 13.
Stiller
CA. International variations in the
incidence of childhood carcinomas. Cancer Epidemiol Biomarkers Prev
1994;3:305-10. 14.
Bortolotti
F. Chronic viral hepatitis in childhood. Baillier,s Clin Gastroenterol
1996;10:185-206. |
15.
Ojanguren
I, Castella E, Ariza et al. Liver cell atypia: A comparative study in
cirrhosis with and without hepatocellular carcinoma.
Histopathology 1997;30:106-12. 16.
Lin
HH, Shyu WC, Chen GL et al. DNA
measurement in chronic hepatitis, cirrhosis and
hepatocellular carcinoma. Liver 1990;10:313-8. 17.
Tanaka
N, Ohtsuka S, Honda et al. Multiparametric cytometric analysis of hepatocellular
carcinoma and its allied lesions combining DNA
ploidy analysis with morphometry using DAPI/HP
double staining. Gan To Ryoho 1995;22:197-204 (English abstract). 18.
Dangwal
TR, Aggarwal V, Malhgtra V et al. Clinical spectrum of chronic liver
disease in north Indian children. Trop Gastroenterol
1997;18:174- 6. 19.
Abdel
Aziz F, Habib M, Mohamed MK et al. Hepatitis C virus (HCV) infection in a community in the Nile Delta: Population
description and HCV prevalence. Hepatology
2000;32:111-5. 20.
Okuyama
K. Primary liver carcinoma associated with biliary cirrhosis due to
congenital biliary atresia. J Pediatr 1965;67:89-93. 21.
Ishak
KG, Glunz PR. Hepatoblastoma and hepatocarcinoma in infancy and childhood: Report of 47 cases. Cancer 1967;20:396-422. 22.
Deiras
MP, Dicus W. Hepatocarcinoma associated with biliary cirrhosis. A case due to
congenital bile duct atresia. Arch Pathol
1968;86:338-41. 23.
Lee
CL, Ko YC. Survival and distribution pattern of childhood liver cancer in Taiwan.
Eur J Cancer 1998;34:2064-7. 24.
Chang
MH. Hepatocellular carcinoma in children. Huis Tsa Chih 1998;39:366-70 (English
abstract). 25.
Chen
JC,Chen CC,Chen WJ et al. Hepatocellular carcinoma in children: Clinical review
and comparisson with adult cases. J Pediatr Surg 1998;33:1350-4. 26.
Ronacalli
M, Borzio M, Brando B et al. Abnormal DNA content in liver cell dysplasia: A
flow-cytometric study. Int J Cancer 1989;44:204-7. 27.
Blum
HE. Does hepatitis C virus causes
hepatocellular carcinoma? Hepatology 1994;19:251-8. 28.
Trevisani
F, D,Intino PE, Carceni P et al. Etiologic factors and clinical
presentation of hepatocellular carcinoma: Differences between
cirrhotic and noncirrhotic Italian patients.
Cancer 1995;75:2220-32. 29.
Esteban
R. Risk of hepatitis B in infancy and childhood. Vaccine 1995;13:535-6. 30.
Viviani
S, Jack A, Bah E, Montesano R. Hepatocellular carcinoma, a preventable
cancer. Epidemiol Prev 1997;21:129-36. |
31.
Rahman
SM, Itakura H, Motoda A. Morphometry in histopathology. An image analysis
work station for the pathology laboratory. Anal
Quant Cytol Histol 1996;18:471-80. 32.
Rahman
SM, Itakura H. Regenerative pattern of liver cells in primary biliary
cirrhosis, alcoholic cirrhosis, posthepatitic cirrhosis (HBV
related) and hepatocellular carcinoma.
Comparative analysis by computerised morphometry. Pathology Int 1996; 46:267-73. 33.
Badizedegan
K, Jonas MM, Ott MJ et al. Histopathology of the liver in children
with chronic hepatitis C viral infection. Hepatology 1998;28:1416-23. 34.
Coire
CI, Qizlibash AH, Castelli MF. Hepatic
adenomata in type Ia glycogen storage disease.
Arch Pathol Med 1987;111:166-9. 35.
Ugarte
N, Gonzales G, Grussi F. Hepatoma in siblings with progressive familial cholestatic
cirrhosis of childhood. Am J Clin Pathol
1981;76:172-7. 36.
Rabinowitz
M, Imperial JC, Schade RR et al. Hepatocellular carcinoma in Alagille
sydnroma: A family study. J Pediatr Gastroenterol
Nutr 1989;8:26-30. 37.
Quillin
SP, Brink JA. Hepatoma complicating Byler disease. AJR 1992;159:432-3. 38.
Moore
L, Bourne AJ, Moore DJ et al. Hepatocellular carcinoma following
neonatal hepatitis. Pediatr Pathol Lab Med
1997;17:601- 10. 39.
Lin
XZ, Horng MH, Sun YN et al. Computer
morphometry for quantitative measurement of liver fibrosis: Comparisson with Knodell,s
score, colorimetry, and conventional description reports. J Gastroenterol Hepatol 1998;13:75-80. 40.
Pilette
C, Rousselet MC, Bedossa P et al. Histopathological evaluation of liver
fibrosis quantitative image analysis vs
semiquantitative scores. Comaprisson with serum markers.
J Hepatol 1998;28:439-46. 41.
Zaitoun
AM, Al Mardini H, Awad S, Ukabam S, Makadisi S, Recard CO. Quantitative assessment of fibrosis and steatosis in liver
biopsies from patients with chronic hepatitis. J Clin
Pathol 2001;54:461-5. 42.
Berman
MA, Burnham JA, Sheahan DG. Fibrolamelar carcinoma of the liver: An
immunohistochemical study of ninteen cases and a revuew of the literature. Hum Pathol
1988;19:784-94. |