|
|
Alimentary
tract and pancreas Alimentarni
trakt i pankreas |
||
|
1Miodrag @ivi}, 2Vuka Kati}, 1Dragoljub Popovi}, 3Aleksandar Nagorni, 4Slobodan Trenki}, 4Neboj{a \or|evi}, 2Vesna @ivkovi} , 2Katarina Kati}, 5Sa{a Grgov, 6Marica Ota{evi}. Clinic for Otorhinolaryngology, Clinical Center Niš, 2 Institute of Pathology, Medical Faculty of Niš, 3 Clinic for Gastroenterology and Hepatology, Clinical Center Niš, 4 Surgical Clinic, Clinical Center Niš, 5 Department of Gastroenterology, Health Centre of Leskovac, 6 Institute of Microbiology, Medical Faculty of Niš. . |
ARCH
GASTROENTEROHEPATOL 2003; 22 (No 1 - 2): 12 – 17 Histological,
histochemical and
clinical features of Barrett's
oesophagus Histolo{ke,
histohemijske i klini~ke odlike Barrett-ovog
jednjaka (accepted
April 4th 2003 ) |
||
|
Key Words: Barrett's oesophagus, histopathology, epithelial mucins, genetic..
|
Abstract Barrett's
oesophagus refers to an acquired change charcterised by replacement of the
normal epithelium of
the lower oesophagus by a columnar epithelium and ccuring in subjects with
gastroesophageal reflux disease.
The mucosa represents a complex mixture of cell types and architectural
patterns found in the stomach
and small intestine.Gastric metaplasia (also termed cardiac type) comprises
surface and foveolar epithelium
lined by a columnar mucous cells and cardiac or pyloric-type mucous
glands.Chief and parietal
cells may be present but are rarely conspicuous. Intestinal metaplasia is
typically incomplete, comprising
intestinal goblet cells and gastric foveolar type columnar mucous cells. The
association of Barrett's
metaplasia and oesophageal adenocarcinoma has long been recognized and cancer
surveillance by
regular endoscopic examination has been advocated by many investigators.
Progression of Barrett's epithelium
to dysplasia and malignancy is usually accompanied by downregulation of
secretory mucins MUC2,
MUC5AC and MUC6, similar to cancer of stomach and colon. Membrane bound
mucins MUC1
and MUC4 may show upregulation, whereas the membrane bound mucin MUC3 is
downregulated. Early
premalignant clones produce biological and genetic heterogeneity as seen by
multiple p53 mutations,
p16 mutations, aneuploidy, and abnormal methylation resulting in stepwise
changes in differentiation, proliferation
and apoptosis, allowing disease progression under selective pressure. Exploitation
of these molecular events may lead to a more appropriate diagnosis and
understanding of these
lesions in the future. Sa`etak Barrett-ov
jednjak je ste~ena promena koju odlikuje zamena normalnog epitela donjeg dela
jednjaka cilindri} nim
epitelom i koja se karakteristi~no javlja u osoba sa gastroezofagusnim
refluksom. Sluzoko`a prestavlja
slo`enu me{avinu }elijskih tipova i gradje prisutne u `eludcu u tankom crevu.
Gastri~na metaplazija (takodje
nazvana kardijalinim tipom) ~ini pokrovni i foveolarni epitel oblo`en
cilindri~nim mukusnim }elijama
i kardijalnim ili pilori~nim tipom `lezda. Glavne i parijetalne }elije mogu
da budu prisutne, ali su
retko upadljive. Intestinalna metapolazija je karakteristi~no inkompletna i
gradjena je od intestinalnh peharastih
}elija, mukusnih i gastri~nih }elija foveolarnog tipa. Udru`enost metaplazije
i adenokarcinoma jednjaka
je odavno propoznata, a otkrivanje karcinoma tokom redovnih endoskopskih
kontrola zastupaju mnogi
istra`iva~i. Progresija Barrett-ovog epitela u displaziju i malignitet je obi~no
pra}ena smanjenim lu~enjem
sekretornih mucina tipa MUC2, MUC5AC i MUC6, sli~no karcinomu `eludca i
kolona. Mucini koji su
vezani za membranu tipa MUC1 i MUC4 mogu da poka`u hipersekreciju dok su
membranski mucini tipa
MUC3 u hiposkreciji. Rani premaligni klonvi stvaraju biolo{ku i genetsku
heterogenost multiplih p53
mutacija, p16 mutacija, aneuploidiju i abnormalnu metilaciju dovode}i do
stepeni~astih promena diferencijacije, proliferacije
i apoptoze, i dozvoljavaju}i napredovanje bolesti pod selektivnim pritiskom. Poznavanje
ovih molekularnih zbivanja i njihovo kori{}enje u praksi mo`e da dovede do
rane dijagnoze i ve~eg
razumevanja ovih promena u budu}nosti. |
||
|
Kljucne reci: funkcionalna dispepsija, podgrupe, Helicobacter pylori. |
|||
|
|
History
of the Columnar-Lined Oesophagus Barrett
was not the first to describe the columnar-lined oesophagus.
In 1906, Tileston, a pathologist, reported several patients
who had ²peptic ulcer of the oesophagus² and noted
the close resemblance of the mucous membrane about
the ulcer to that normally found in the stomach (1). In
1950, an influential British surgeon named Norman Barrett
published a report in which he defined the oesophagus as
²that part of the foregut, distal to the cricopharingeas sphincter,
which is lined by squamous epithelium² (2).
Today, an oesophagus lined extensively by columnar epithelium
is called, ironically, Barrett's esophagus (BE) (3).
The condition is associated both with gastroesophageal reflux
disease (GERD) and oesophageal adenocarcinoma. Barrett's
oesophagus This
term refers to an acquired change characterised by replacement
of the squamous epithelium of the lower oesophagus
by columnar epithelium and occuring in subjects with
GERD. The mucosa represents a complex mixture of
cell types and architectural patterns found in the stomach
and small intestine (4). While
the various types of epithelium lining BE may not
be arranged in definitive zonal distribution as thought originally,
it is nevertheless possible to observe discrete areas
of gastric metaplasia and intestinal metaplasia. Gastric
metaplasia (also termed cardiac type)comprises surface
and foveolar epithelium lined by columnar mucous cells
and cardiac or pyloric-type mucous glands. Chief and parietal
cells may be present but are rarely conspicious. Intestinal
metaplasia is typically incomplete, comprising intestinal
goblet cells and gastric foveolar type columnar mucous
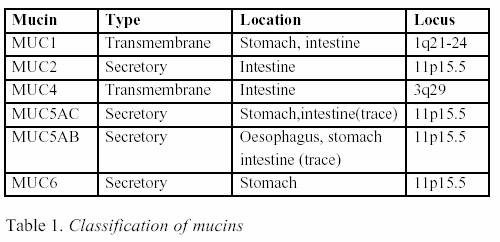
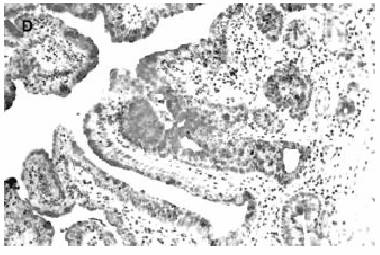
cells (also termed specialised type epithelium). Apsorptive
cells and Paneth cells are inconspicuous. Intestinalised
Barrett's epithelium is more likely to show a villous
architecture (Fig.1) and is recognised as the precursor od
dysplasia and adenocarcinoma (5). Although some restrict
the diagnosis of Barrett's oesophagus to cases with intestinal
metaplasia, gastric or cardiac metaplasia is an abnormal
finding and a marker for GERD (6). Barrett's
mucosa is recognised in HE stained sections not
only by presence of goblet cells but by distinctive architectural changes
including villosity and crypt architectural abnormalities
(loss of parallelism, tortuosity, branching and varying
degrees of atrophy) (Fig 1). Squamous islands and ducts
of submucosal oesophageal glands confirm the site of biopsied
columnar epithelium.The lamina propria may be replaced
by fibromuscular tissue and splitting of the muscular mucosae
into two layers is often obeserved in surgical specimens.
Barrett's mucosa is therefore more than a simple epithelial
change dominated by goblet cell metaplasia. Goblet
cells in gastroesophageal biopsies are not pathognomonic for
the condition. These cells may be seen in intestinal metaplasia
of the cardia associated with Helicobacter pylori
gastritis. In this situation,however, intestinal metaplasia is
likely to be complete (7). Factors
predisposing for the development of BE and subsequent
adenocarcinoma in patients with GERD include a
markedly increased oesophageal exposure time to refluxed
gastric and duodenal contents due to a defective barrier
function of the lower oesophageal sphincter and ineffective
clearance function of the tubular oesophagus. Experimental
and clinical data indicate that combined oesophageal
exposure to gastric acid and duodenal contents (bile
acids and pancreatic enzymes) appears to be more detrimental
than isolated exposure to gastric juice of duodenal contents
alone. Combined reflux is thought to increase
cancer risk by promoting cellular proliferation, and by
exposing the oesophageal epithelium to potentially genotoxic
gastric and intestinal contents, e.g. nitrosamines (8). Symptoms
and signs BE
as the precursor of most adenocarcinomas is clinically silent
in up to 90% of cases. The symptomatology of BE,
when present, is that of gastro-oesophageal reflux. This is
the condition where the early stages of neoplasia (intraepithelial and
intramucosal neoplasia)should be sought (9). Endoscopy The
endoscopic analysis of the squamocolumnar junction aims
at the detection of columnar metaplasia in the distal oesophagus.
At endoscopy, the squamocolumnar junction (Z-line)
is in the thorax, just above the narrowed passage across
the diaphragm. In the lenght of the columnar lining
in this distal oesophageal segment is more than 3cm, it
is termed a long type of Barrett metaplasia. When the length
is less than 3 cm, it is a short type. Single or multiple finger-like
(1-3 cm) protrusions of columnar mucosa are classified
as short type.In patients with short segment BE, the
risk for developing adenocarcinoma is reported to be lower
compared to those with long segment BE (10). As BE is
restricted to cases with histologically confirmed intestinal metaplasia,
adequate tissue sampling is required. Histopathology Barrett
epithelium is characterized by two different types
of cells, i.e. goblet cells and columnar cells, and has also
been termed 'specialized', 'distinctive' or Barrett metaplasia. The
goblet cells stain positively with Alcian blue at low
pH (2.5). The metaplastic epithelium has a flat or villiform surface,
and is identical to gastric intestinal metaplasia of
the incomplete type (type II or III) (Fig.1). Rarely, foci
of complete intestinal metaplasia (type I) with absorptive cells
and Paneth cells may be found. The mucous glands
beneath the surface epithelium and pits may also contain
metaplatic epithelium. Recent studies suggest that the
columnar metaplasia originates from multipotential cells
located in intrinsic oesophageal glands (11). Intraepithelial
neoplasia in Barrett oesophagus Macroscopy
: intraepithelial neoplasia generally has no distinctive
gross features, and is detected by systematic sampling
of a flat Barrett mucosa. The area involved is variable,
and the presence of multiple dysplastic foci is common
(12). In some cases, intraepithelial neoplasia presents as
one or several nodular masses resembling sessile adenomas.
Rare dysplastic lesions have been considered true
adenomas, with an expanding but localised growth resulting
in a well demarcated interface with the surrounding tissue. Microscopy:
abnormal proliferation and differentiation typify
epithelial dysplasia.Normal oesophageal squamous epithelial
cells divide slowly in the basal layer, proliferate suprabasally,
and mature towards the luminal surface (13). In
Barrett's mucosa, despite its partially intestinal phenotype, proliferation
and differentiation patterns resemble gastric
mucosa, with minimal proliferation in a crypt zone beneath
the mucosal surface and differentiation into deep glands
and characteristic cell populations on the mucosal surface
(in normal small intestine,stem cells in the crypts of
Lieberkuhn feed a proliferative compartment from which
differentiating enterocytes and goblet cells migrate to
the villi,while Paneth cells migrate basally) (13). Proliferation
and differentiation compartments break down in
dysplastic epithelia. ìDysplasticì
cells
adjacent to an invasive
carcinoma probably represent the neoplastic clone from
which the carcinoma emerged. Dysplasia alone implies
an increased cancer risk, and motivates eradication or
increased intensity of surveillance (14). Epithelial
atypia in Barrett mucosa is usually assessed according
to the system devised for atypia in ulcerative colitis,namely:
negative, positive or indefinite for intraepithelial neoplasia. Negative
for intraepithelial neoplasia:usually, the lamina propria
of Barrett mucosa contains a mild accompanying inflammatory
infiltrate of mononuclear cells.There may
be mild reactive changes with enlarged, hyperchromatic nuclei,
prominence of nucleoli, and occasional mild stratification
in the lower portion of the glands.However, towards
the surface there is maturation of the epithelium with
few or no abnormalities.These changes meet the criteria of
atypia negative for intraepithelial neoplasia, and can
usually be separated from low-grade intraepithelial neoplasia
(15). Atypia
indefinite for intraepithelial neoplasia:one of the major
challenges for the pathologist in Barrett oesophagus is
the differentiation of intraepithelial neoplasia from reactive
or regenerative epithelial changes.This is particularly difficult,
sometimes impossible, if erosions or ulcerations are
present (16). In areas adjacent to erosions and ulcerations,
the metaplastic epithelium may display villiform hyperplasia
of the surface foveolae with cytological atypia
and architectural disturbances. These abnormalities are
usually milder than those observed in intraepithelial neoplasia.
There is a normal expansion of the basal replication zone
in regenerative epithelium versus intraepithelial neoplasia,
where the proliferation shifts to more superficial portions
of the gland. If there is doubt as to whether reactive
and regenerative changes or intraepithelial neoplasia is
present in a biopsy, the category atypia indefinite for intraepithelial
neoplasia is appropriate and a repeat biopsy after
reflux control by medical acid supression or antireflux therapy
is indicated. Low-grade
and high grade intraepithelial neoplasia:intraepithelial
neoplasia in Barrett metaplastic mucosa
is defined as a neoplastic process limited to the epithelium
(17). Its prevalence in Barrett mucosa is approximately
10%, and it develops only in the intestinal type
metaplastic epithelium. Cytological abnormalities typically
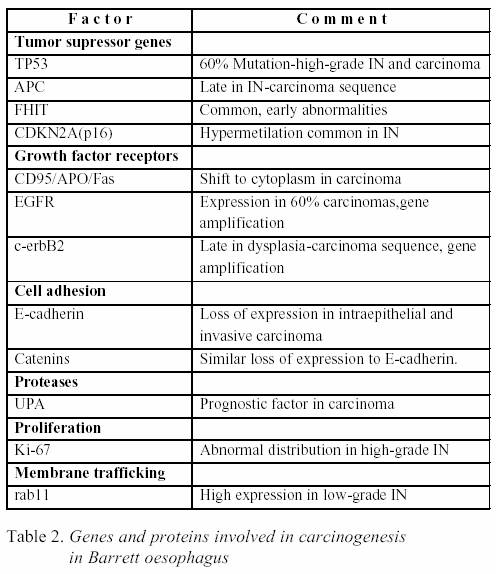
extend to the surface of the mucosa. In low-grade intraepithelial
neoplasia, there is decreased mucus secretion, nuclear
pseudostratification confined to the lower half of the
glandular epithelium, occasional mitosis, mild pleomorphism, and
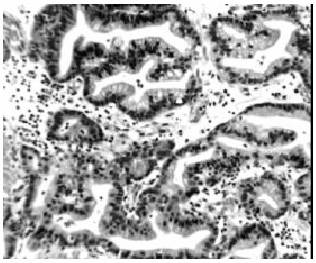
minimal architectural changes (Fig 2,left). High-grade
intraepithelial neoplasia shows marked pleomorphism and
decrease of mucus secretion, frequent mitosis, nuclear
stratification extending to the upper part of the cells
and glands, and marked architectural aberrations ( Fig 2,
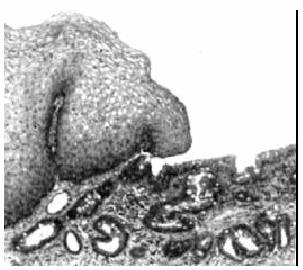
right). The most severe architectural changes consist of a
cribriform pattern that is a feature of high-grade intraepithelial neoplasia
as a long as the basement membrane of the
neoplastic glands has not been disrupted (Fig 3).The diagnostic
reproducibility of intraepithelial neoplasia is far from
perfect; significant interobserved variation exists (18). Mucin
expression in Barret's oesophagus Mucins
are glycoproteins containing up to 85% carbohydrate. The
oligosaccharide chains contain 2 to 12 sugars. Each
chain consists of a core region, a backbone and a peripheral
blood group-type structure.Two sugars may be added
as side-groups to the chain: sialic acid and fucose. Carbohydrate
component may be studied histochemically by
meand of traditional mucin staining, lectin binding and immunohistochemistry.
Many of the techniques are complex, involve
digestion or blockade and have generally resulted
in a literature that is difficult to disentagle. The
polypeptide component consists of a central domain
made up of repeating amino acid sequences which carry
the carbohydrate chains and peripheral domains that are
less heavily glycosylated. In MUC2, for example, which
is the dominant mucin of intestinal goblet cells, the length
of each repeat is 23 amino acids and these are repeated
between 51 and 150 times (19). The colorectal expresses
MUC1 which is a structural glycoprotein with intracytoplasmic
transmembrane and extracellular domains.
It is expressed at the apical pole of epithelial cells,
particularly within the crypt base of normal colorectum. Immunogold
labelling at the EM level shows that MUC1
is produced by goblet cells.MUC 3 and MUC4 appear
to be most abundantly expressed by columnar cells of
normal colorectum. Studies
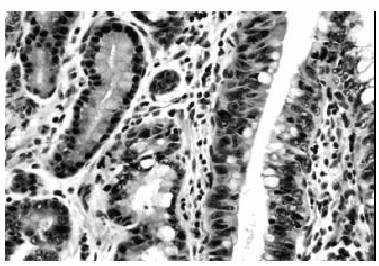
of mucin gene expression in Barrett's oesophagus reveal
the expression of gastric mucins MUC5AC (Fig 4)
and MUC6 as well as intestinal mucins (MUC2,MUC3 and
MUC4) by 'intestinalised' epithelium (20).This findings confirm
the incomplete nature of intestinalisation in Barrett's
oesophagus. A similar mucin pattern is observed in
incomplete intestinal metaplasia of gastric mucosa (21) However,
whereas MUC5AC appears to be restricted to columnar
cells in Barrett's oesophagus, it is expressed by both
goblet cells and columnar cells in incomplete intestinal metaplasia
of the stomach (22). As in incomplete intestinal
metaplasia of gastric mucosa, subtypes of Barrett's
epithelium secreting sulphomucin have been associated
with neoplastic progression. This finding occurs with
high frequency in Barrett's epithelium and has not translated
into a practical marker of increased risk. It
is unclear whether Barrett's mucosa develops as an upward
extension of a metaplastic gastric mucosa or represents a
transformation of stem cells within the ducts of oesophageal
mucous glands (23).The presence of MUC5B in
oesophageal glands but not in Barrett's oesophagus fits the
first hypothesis (22).The finding of cytokeratin (CK 7) within
both Barrett's epithelium and oesophageal glands but
not to the same extent in intestinal metaplasia of gastric mucosa
supports the second hypothesis. The possibility that
reflux injury in the lower oesophagus could be caused
by defective oesophageal mucin production has been
considered (24). The main secretory mucin MUC5B is
produced by the submucous oesophageal glands but membrane
bound mucins (MUC1 and MUC4) are expressed
by oesophageal squamous epithelium and may have
an important cytoprotective role.The mixed gastric and
intestinal phenotype characterising intestinalised mucosa
could represent an adaptation to cell injury mediated by
the combination of gastric acid and bile (22). A cytoprotective
role is supported by the demonstration of expression
of trefoil peptides TFF1 and TFF2 by Barrett's epithelium. Progression
of Barrett's epithelium to dysplasia and malignancy
is usually accompanied by downregulation of secretory
mucins MUC2, MUC5AC and MUC6, similar to cancer
of stomach and colon (25).The other authors have reported
that Barrett's metaplasia expressed MUC2 (an intestinal
mucin), but MUC1 was consistenly absent (26). Neither
MUC1 or MUC2 were expressed in the dysplastic epithelium
whether in its form or when associated with carcinomas.The
lack of MUC1 in dysplastic epithelium and
its expression in carcinoma could be utilyzed as a marker
which could differentiate dysplasia from carcinoma in
mucosal biopsies (26). Furthermore, expression of MUC1
in advanced stage oesophageal cancers suggests an unfavourable
prognosis. Membrane
bound mucins MUC1 and MUC4 may show
upregulation, as in the other regions of the gastrointestinal tract,
whereas the membrane bound mucin MUC3 is
downregulated (Table 1) (19).
Immunohistochemistry Minichromosome
maintenance (Mcm) proteins are essential
for eukaryotic DNA replication, and their expression implies
potential for cell proliferation. xpression is dysregulated
in dysplastic states, but data for oesophageal squamous
mucosa and Barrett's mucosa have not been published (13). Immunostaining
with the Mcm2 antibody yielded predominantly nuclear
staining. The Mcm5 antibody stains nuclei
but also cell membranes in glandular mucosae and tumours.
Qualitatively, nuclear staining is similar with the two
antibodies. Ki-67 staining is purely nuclear. In
non-dysplastic squamous epithelium and Barrett's mucosa,
strong Mcm2, Mcm5, and Ki-67 staining of most to
all nuclei are present in the expected proliferative transit compartment-that
is, the suprabasal compartment of squamous
epithelium-and in the lower crypt compartments of
Barrett's mucosa. In differentiated compartments-that is, the
surface of squamous epithelium and Barrett's mucosaand in
the small differentiated deep glands of Barrett's mucosa,
expression is downregulated. In dysplastic squamous epithelium
and dysplastic Barrett's mucosa there is persistence
of Mcm2, Mcm5,and Ki-67 expression in compartment in
which they are normally absent or sparse, especially
towards the surface of squamous epithelium and Barrett's
mucosa. Downregulation of Mcm2 and Mcm5 expression
in the deep(glandular) mucosal compartment of Barrett's
mucosa is also significantly reduced in high grade dysplasia
(13,14). Genetics In Barrett
oesophagus a variety of molecular genetic changes
has been correlated with the metaplasia-dysplasiacarcinoma sequence
(27). Prospective follow-up of lesions biopsied
at endoscopy show that alterations in TP53 and CDKN2A
occur at early stages (28). TP53:
in high-grade intraepithelial neoplasia a prevalence of
TP53 mutations of approximately 60% is found, similar
to adenocarcinoma. Mutation in one allele is often accompanied
by loss of the other (17p13.1). Mutations occur
in diploid cells and precede aneuploidy. The pattern of
mutations differs significantly from that in squamous cell
carcinomas. CDKN2A:
alterations of CDKN2A, a locus on 9p21 encoding
two distinct tumour suppressors, p16 and p19 include
hypermethylation of the p16 promotor and, more rarely,
mutations and LOH (29). FHIT:
among other early changes in the premalignant stages
of metaplasia are alterations of the transcripts of FHIT,
a presumptive tumour supressor gene spanning the common
fragile site FRA3B (30). LOH
and gene amplification: a number of other loci are altered
relatively late during the development of adenocarcinoma, with
no obligate sequence of events. Prevalent changes
include LOH on chromosomes 4 (long arm) and 5 (several
foci including APC) and amplification of ErbB2 (31). Phenotypic
changes in Barrett oesophagus includeexpansion of the Ki-67 proliferation
compartment correlating with
the degree of intraepithelial neoplasia. Molecules involved
in membrane trafficking such as rabll have reported
to be specific for the loss of polarity seen in lowgrade intraepithelial
neoplasia. In invasive carcinoma, reduced
expression of cadherin/catenin complex and increased
expression of various proteases are detectable Table
2) (31). Non-neoplastic Barrett oesophagus expresses the
MUC2 but not the MUC1 mucin gene product, whereas
neither is expressed in intraepithelial neoplasia in Barrett
oesophagus (31). Invasive lesions exhibit variable expression
of MUC1 and MUC2.
CONCLUDING
REMARCS Although
the origin of BE is a matter of conjecture, one current
theory holds that the stem cells of squamous mucosa or
associated glandular ducts undergo altered differentiation, producing
both microvilli and intercellular ridges, and express unique
glandular phenotypes distinct from adjacent mucosal gastric
stem cells.This Barrett's metaplastic lineage may give rise
to Paneth cells and neuroendocrine cells in addition to gastric
and intestinal cells and is therefore pluripotent (32). Curent
theory indicates that these cells give rise to intestinaltype metaplasia.
However, skeptics argue that gastric type, fundic
type and sebaceous gland metaplasia (33) are also descernible
and that these metaplasia may more accurately be refered
to as a mosaic, although a convincing paradigm is lacking
(24,25,31,32). The reason behind this heterogeneity of metaplastic
phenotypes is unclear but the proportion of each has
been attributed in part to the composition of the refluxate (environment).
Phenotypic heterogeneity may also be controlled genetically
because clonal divergence in chromosomes 5,8,9,12,17,
and 18 in nondysplastic Barrett's cells can also be identified
(34). The
location and composition of the proliferative compartment in
the crypts of the metaplastic epithelium are not as well
defined as in columnar lined epithelium of the stomach (32).
Interestingly, the degree of which differentiation occurs varies
considerably.BE that appears in childhood differs from the
adult variety in that intestinal mucins and cytokeratins are not
present. The adult variety also has an inflammatory cell infiltrate
and may have Helicobacter-like
organisms, both of which
are less common in juvenile metaplasia. Barrett's intestinal
phenotype has higher proliferative indices; this is associated
with altered expression of multiple growth factors and
inducible nitric oxide synthase and cyclooxygenase-2. Dysplastic
cells may have proliferative controls that are relaxed
or uncoupled from the appropriate regulatory clues. In part
this may be a result of altered expression of cytokines and growth
factors, although the acquisition of genomic alterations of
cell-cycle-associated genes also occurs.These cell cycle
genes include increased cyclin D1 expression(chromosome 11q13),
hypermethylated or mutated p16 (chromosome 9p21),
and mobilization of cells from G0 to G1, with subsequent accumulation
in the G2 phase.Identification of increased
telomerase RNA in early dysplastic lesions including Barrett's
metaplasia has been reported. P53 mutations occur
in only 1-5% of metaplastic diploid cell populations but are
present in most aneuploid cells, suggesting they are usually not
early events (35,36). Epigenetic alterations in the expression
of growth factors and their receptors, especially of the
epidermal growth factor family, are also associated with these
cell cycle changes in dysplastic Barrett's mucosa. In particular, the
authors beleive that increased expression of TGFalfa,
and its precursor preproTGFalfa, may stimulate epidermal growth
factor receptors in dysplastic cells by autocrine and
paracrine mechanisms, respectively (37). Apoptosis
may also be inhibited late in a proportion of dysplastic
cells that give rise to invasive or metastatic cells (38).
The bcl-2 gene is not overexpressed, as is recognized in colorectal
adenomas, although p53 mutations may affect the proliferation
/apoptosis ratio in the oesophagus (39). In addition, up-regulation
of immunological death factors such as Fas
ligand in the epithelium may not only protect Barrett's dysplastic
cells but also may selectively destroy cytotoxic T cells
by crosslinking Fas (40).
Figure
1. Barrett oesophagus: dysplastic intestinal
lands(right). HE
x 200.
Figure
2. Low-grade intraepithelial neoplasia on the left
and high grade
on the right. HEx300.
Figure
3. Pyloric type of dysplastic metaplasia. HE x 250.
Figure
4. Conspicuous expression of MUC5AC. ABC x 250. Recent
evidence has shown that Barrett metaplasia and dysplasia
may partly involve down-regulation, mutation, or phosphorylation
of cadherin / catenin the adhesion complexes, thereby
increasing free cytosolic catenin.Increased betacatenin levels
have been shown to subsequently aggregate with
transcription factors in the nucleus, facilitating epitheliomesenchymal transition
and increased c-myc expression (41). |
||
|
|
REFERENCES: 1.
Spechler
SJ and Goyal RK. The columnar-lined oesophagus, intestinal metaplasia and
normal Barrett.Gastroenterology 1996; 110 :
614-21. 2.
Barrett
NR. Chronic peptic ulcer of the oesophagus and 'oesophagitis'. Br J Surg 1950;
38:175-82. 3.
Spechler
SJ. Barrett's esophagus.Semin Oncol 1994; 21:431-7. 4.
Jass
JR, Roberton AM. Colorectal mucin histochemistry in health and disease: A critical
review. Pathol. Int. 1995; 44:487-504. 5.
Reid
BJ, Weinstein WM. Barrett's oesophagus and adenocarcinoma. Annu. Rev. Med. 1987;
38: 477- 92. 6.
Chandrasoma
PT, Lokuhetty DM, Demeester TR. Definition of histopathologic changes
in gastroesophageal reflux disease. Am J Surg Pathol 2000; 24: 344-51. 7.
Voutlainem
M, Farkkila M, Juhola M, Meckin JP, Sipponen P. Complete and incomplete intestinal metaplasia at the oesophagogastric
junction: prevalences and asociations with
endoscopic erosive oesophagitis and gastritis. Gut 1990;
45:644- 8. 8.
Stein
HJ, Kauer WK, Feussner H, Siewert JR. Bile reflux in benign and malignant
Barrett's oesophagus: effect of medical acid
supression and nissen fundoplication. J Gastrointest
Surg 1998 ; 2: 333-41. 9.
Lambert
R. The role of endoscopy in the prevention of oesophagogastric cancer.Endoscopy
1999; 31: 180-99. 10.
Sharma
P, Morales TG, Bhattacharyya A, Garewal HS, Sampliner RE. Dysplasia in shortsegment Barrett's oesophagus: a prospective 3- year follow-up. Am J Gastroenterology
1997; 92: 2012-6. 11.
Ormsby
AH, Goldblum JR, Rice TW, Richter JF, Falk GW. Cytokeratin subsets can realibly distinguish Barrett's esophagus from intestinal
metaplasia of the stomach. Hum Pathol 1999; 30:
288- 94. 12.
Cameron
AJ, Carpenter HA. Barrett's esophagus, high-grade dysplasia, and early
adenocarcinoma: a pathological study. Am J Gastroenterol
1997; 92: 586- 91. 13.
Going
JJ, Keith WN, Neilson L, Stoeber K, Stuart RC and Williams GH. Aberrant expression of minichromosome maintenance proteins 2
and 5, and Ki-67 in dysplastic squamous
oesophageal epithelium and Barrett's mucosa. Gut
2002; 50: 373-7. 14.
Baak
JPA, ten Kate FJW, Offerhaus GJA, van Lanschot JJ, Meijer GA. Routine morphometrical analysis can improve reproducibility of
dysplasia grade in Barrett's oesophagus
surveillance biopsies. Journal of Clinical Pathology 2002; 55: 910-6. |
15.
Antonioli
DA, Wang HH. Morphology of Barrett's oesophagus and Barret's associated
dysplasia and adenocarcinoma. Gastroenterol Clin
North Am 1997; 26: 495-506. 16.
Levine
AJ, Appelman HD. Atlas of tumor pathology. Tumors of oesophagus and stomach 1966; AFIP:Washington DC. 17.
Riddell
RH, Goldman H, Ransohoff DF, Correa P, Hamilton SR, Morson BC. Dysplasia in inflammatory bowel disease . Hum Pathol 1983; 14: 931-68. 18.
Reid
BJ, Haggitt RC, Rubin CE, Roth G, Surawicy CM, Van-Belle G, Lewin K,
Weinstein WM, Goldman H. Observer variation in the diagnosis of dysplasia in Barrett's oesophagus.
Hum Pathol 1988; 19: 166-78. 19.
Jass
JR, Walch MD. Altered mucin expression in the gastrointestinal tract: a review. J
Cell Mol Med 2001; 5: 327-51. 20.
Guillem
O, Billeret V, Buisine MP. Mucin gene expression and cell differentiation in
human normal ,premalignant and malignant esophagus.
A retrospective study. Int J Cancer 2000:
88: 856-61. 21.
Filipe
MI. Mucins in the human gastrointestinal epithelium: a review.Invest Cell Pathol
1979; 2: 195-216. 22.
Arul
GS, Moorghen M, Myerscough N et al. Mucin gene expression in Barrett's
oesophagus: an in situ hybridisation and
immunohistochemical study. Gut 2000; 47: 753-61. 23.
Li
H, Walsh WH, O'Dowd G et al. Mechanisms
of columnar metaplasia and squamous regeneration in experimental Barrett's
oesophagus.Endoscopy 1994; 2: 121-6. 24.
Namiot
Z, Sarosiek J, Rourk RM, Hetzel DP, McCallum RW. Human esophageal secretion: mucosal response to luminal acid and
pepsin. Gastroenterology 1994; 106: 973-81. 25.
Jass
JR. Mucin histochemistry of the columnar epithelium .Virchows Arch 1981; 414:
359-63. 26.
Chinyama
CN, Marshall RE, Owen WJ, Masson RC, Kothari D, Wilkinson ML,
Sanderson JD. Expression of MUC1 and MUC2 mucin gene products in Barrett's metaplasia,
dysplasia and adenocarcinoma: an immunopathological
study with clinical correlation.
Histopathology 1999; 35:517-24. 27.
Werner
M, Mueller J, Walch A, Hofler H. The molecular pathology of Barrett's
esophagus. Histol Histopathol 1999; 14: 553-9. 28.
Barrett
MT, Sanchez CA, Prevo LJ, Wong DJ, Galipeau C. Evolution of neoplastic cell lineages in Barrett esophagus. Nat Genet 1999;
22: 106-9. 29.
Klump
B, Hsiech CJ, Holyman K, Gregor M, Porschen R. Hypermethylation of the CDKN2/p16 promoter during neoplastic
progression in Barrett's oesophagus.
Gastroenterology 1998; 115: 1381-6. |
30.
Michael
D, Beer DG, Wilke CW, Miller DE, Glover TW. Frequent deletions of FHIT and FRA3B in Barrett's metaplasia and
esophageal adenocarcinomas. Oncogene 1997; 15:
1653-9. 31.
Jankowski
JA, Wright NA, Meltzer SJ, Tradafilopoulos G, Geboes K,
Gasson AG, Kerr D, Young LS. Molecular evolution of the metaplasia- dysplasia-adenocarcinoma sequence in
the esophagus. American Journal of
Pathology 1999; 154: 965-73. 32.
Jankowski
JA, Wright NA. Epithelial stem cells in the gastrointestinal tract:structure,
function and adaptation. Sem Cell Biol 1992; 3:
445-56. 33.
Kushima
R, von Hinuber G, Lessel W, Stolte M. Sebaceous gland metaplasia in
cardiac-type mucosa of the oesophago-gastric
junction. Virchows Arch 1996; 428: 297-9. 34.
Wu
TT, Watanabe T, Heitmiller R, Zahurak M, Hamilton SR. Genetic alteration in Barrett esophagus and adenocarcinomas of the esophagus
and esophagogastric junction region.Am J
Pathol 1998; 153: 287-94. 35.
Coppola
D, Falcone R, Livingston S, Karl R, Nicosia S, Cacho CM. Cyclin D1 correlates with degrees of dysplasia in Barrett's
esophagus. Lab Invest 1997; 76: 298-302. 36.
Neshat
H, Sanchez CA, Galipeau PC, Blount PL, Levine DS, Joslyn G, Reid BJ: p53 mutations in Barrett's adenocarcinoma and high grade
dysplasia. Gastroenterology 1994; 106: 1589-95. 37.
Brito
M, Filipe MI, Linehan J, Jankowski J. Transforming growth factor alfa
expression in gastro-esophageal tumorogenesis may
reflect altered processing of the precursor
peptide. Int J Cancer 1995; 60: 27-32. 38.
Katada
N, Hinder RA, Smyrk TC, Hirabayashi N, Perdikis G, Lund RJ. Apoptosis is inhibited early in the dysplasia -carcinoma sequence of
Barrett's esophagus. Arch Surg 1997; 132: 728-32. 39.
Goldblum
JR, Rice TV. Bcl-2 protein expression in Barrett's metaplasia, dysplasia carcinoma sequence. Mod Pathol 1995; 8: 866-9. 40.
Fan
XJ, Crowe SE, Bamford K, Van Houten N, Reyes VE, Ernst PB. Fas-mediated apoptosis of gastric epithelial
cells.Gastroenterology 1997; 110 : A13 41.
Morin
PJ, Sparks AB, Korinek V, Banker N, Clevers H, Vogelstein B,
Kinzler KW. Activation of beta:catenin-Tcf signaling in colon
cancer by mutations in beta-catenin of APC.
Science 1997; 275: 1787-90. |