|
|
Alimentary
tract and pancreas Alimentarni
trakt i pankreas |
||
|
Fenyvesi Attila, Department of Pathology, General Hospital,Health Center " Dr Gero István" Senta, Serbia |
ARCH
GASTROENTEROHEPATOL 2003; 22 (No 1 - 2): 12 – 17 Assessment
of the matrix metalloproteinase-9
(MMP-9) expression
in colorectal cancer:
absence of correlation with
prognosis and clinicopathological factors Procena
ekspresije metaloproteinaze-9 ( MMP-9) u
kolorektalnom karcinomu: otsustvo korelacije sa
prognozom i klinikopatološkim faktorima (accepted May 19st, 2001) |
||
|
Key Words: colorectal carcinoma, matrix metalloproteinase-9 (MMP-9), prognosis. |
Abstract Matrix
metalloproteinases family of endopeptideses, degradative enzymes are
considered to play an important
role in tumor invasion and metastasis. Type IV collagen is a major structural
protein in the basement
membrane and extrecellular matrix, thus, much attention has been focused on
the type IV collagenases,
(gelatinases, matrix metalloproteinase-2 and matrix metalloproteinase-9),
which specifically
degrade the basement membrane. To assess the validity of matrix
metalloproteinase-9 as a
prognostic marker of colorectal carcinoma, we performed immunohistochemical
analysis on tissues from
40 invasive colorectal adenocarcinomas. We investigated the relationship
between clinicopathological features
of colorectal carcinoma and matrix metalloproteinase-9 immunoreactivity of the
tumor cells and their potential relationship with patients prognosis. Matrix
metalloproteinase-9 immunoreactivity
was assesed semiquantitatively at the invasive front of colorectal cancer.
There were
no significant associations between intratumoral matrix metalloproteinase-9
expression with major
clinicopathological factors and with overall survival of patients with
colorectal carcinoma. By univariate
Cox hazard-model stage of disease and histological grade of colorectal
carcinoma were a prognostic
factor, while expression of matrix metalloproteinase-9 did not show
prognostic significance. Acknowledgement: Author wishes to thank Toth Tibor MD, PhD
(Department of Pathology and Clinical Cytology, General Hospital Falun, Sweden) for his kind assistance in immunohistohemical
staining. Sazetak Matriks
metaloproteinaze su degradiraju}i fermenti is familije endopeptidaza, za koje
se pretpostavlja da
igraju zna~ajnu ulogu u procesu tumorskog napredovanju i nastanku metastaza.
Kako je kolagen
tip IV glavni strukturni protein bazalne membrane i ekstra}elijskog matriksa
glavna pa`nja je
usmerena na tip IV kolagenaza (gelatinaze, matriks metaloproteinazu-2 i
matriks metaloproteinazu- 9) koje
specifi~no razgradjuju bazalnu membranu. U cilju procene validnosti
aktivnosti enzima metaloproteinaze-9
kao prognosti~kog pokazatelja kolorektalnog karcinoma, mi smo sproveli imunohistohemijsko
ispitivanje tkiva 40 invazivnih kolrektalnih karcinoma. Ispitivali smo odnos izmedju
klinikopatolo{kih pokazatelja kolorektalnih karcinoma i imunoreaktivnosti
matriks metaloproteinaze- 9 u
tumorskim }elijama kao i mogu}u povezanost sa prognozom obolelih osoba.Imunoreaktivnost
matriks metaloproteinaze-9 je procenjivana semikvantitativno u invazivnom
segmentu kolorektalnog karcinoma. Nije utvrdjena zna~ajna korelacija izmedju
intratumorske ekspresije
matriks metaloproteinaze-9 i klju~nih klinopatolo{kih obele`ja odnosno
pre`vljavanja pacijenata
sa kolorektalnim karcinomom. "Cox-hazad" model procene stepena
uznapredovalnosti bolesti
i histolo{ki stadijum kolorektalnog karcinoma su operiraju}i prognosti{ki
faktori, dok ekspresija
matriks metaloproteinaze-9 nije pokazala prognosti~ki zna~aj. |
||
|
Kljucne
reci: kolorektalni
karcinom, matriks
metaloproteinaza-9 (MMP-9), prognoza. |
|||
|
|
INTRODUCTION The
colorectal carcinoma (CRC) is the most common malignant
tumor of the alimentary tract and second major cause
of cancer-associated morbidity and mortality in Europe
(1). At the time of diagnosis, CRC usually shows extensive
local invasion and metastasis. Although many factors
regulate malignant tumor growth and spread, interactions between
a tumor and its surrounding microenvironment result
in the production of important protein products that
are crucial to each step of tumor progression (2). Degradation
of extracellular matrix (ECM) is essential in physiological
processes and several pathological conditions. Tumor
invasion, metastatsis and angiogenesis require
controlled degradation of ECM and increased expression
of matrix metalloproteinases (MMPs) (3). MMPs
are a family of zinc-dependent endopeptideses, degradative
enzymes with clear links to malignancy. These enzymes
are associated with tumor cell invasion of the basement
membrane (BM) and stroma, tumor-induced angiogenesis,
blood vessel penetration and metastasis (4). Currently,
this family of MMPs has at least 17 different members
in humans, collectively capable of degrading almost
all ECM components. According to their structures and
substrate specificities, MMPs can be classified into subgroups
of collagenases, gelatinases, stromelysins, membrane-type
MMPs and other MMPs (5). The degradative ability
of tumor can be through both enzymatic activity of
the tumor cell and through cellular components of the
tumor stroma (3). The expression of MMPs in tumors is
regulated in paracrine manner by growth factors and cytokines
secreted in continuous cross talk processes between
tumor, stromal and inflammatory cells during the invasion
(5). Type IV collagen is a major structural protein in
the BM and ECM (6), thus, much attention has been focused
on the type IV collagenases, (gelatinases A and B, MMP-2
and MMP-9), which specifically degrade the BM (3,5).
MMP-9 is considered to be one of the key enzymes involved
in tumor invasion and metastasis (7). Several recent
studies revealed higher expression of MMP-9 in: agressive
tumor cell lines of human lung carcinoma (8), advanced
stage of melanoma (9), human giant cell tumors of
bone (10). There are many literature data about extensive proteolytic
activity in CRC (11) and its association with
stage, grade of tumor and patients prognosis (12-14). The
aims of this study were investigated by immunohistochemistry the
expression of MMP-9 and their potential relationship
with patients prognosis, metastatic behavior of
tumor and classical clinicopathological prognostic factors
of CRC. PATIENTS
AND METHODS The
study population was composed of 40 patients underwent
curative resection of CRC at the Department of Surgery
of General Hospital Senta from 1990 to 1995. None
of them had received chemotherapy or radiation therapy before
surgery. The operation were standard colon or rectum
resection with regional lymph node dissection. The
tumors were typed and graded according to the criteria of
the World Health Organization classification (15). The
extent of the tumor invasion and presence of metastasis were
based on the Astler-Coller modification of the Dukes
classification system (16). Patients died within 30 days
after surgery were excluded. Only patients classified as
stage C received postoperative chemotherapy using 5- fluorouracil
and leucovorin. Slides
were obtained from the invasive front of tumor (17).
Three-micrometar-thick sections from the selected original
paraffin blocks were cut and rehydrated as usual. Immunohistochemical
staining was carried out in the Ventana ES automated Immunohistochemistry System (Ventana Medical System Inc., Tucson, AZ, USA) using original Ventana reagents, with the exception of the primary antibodies, MMP-9 (Novocastra, lyophilised monoclonal NCL-MMP-9; Novocastra Laboratories Ltd., Newcastle upon Tyne, UK). Antigen retrieval was performed in 0.1 M citric acid puffer, pH 7,3, with heating from room
temepature to 97oC. Primary
antibody MMP-9 was used at the dilution of 1:25 with reaction time of 32 minutes. The slides were weakly counterstained with hematoxylin and were mounted routinely. The intratumoral expression of MMP-9 was analyzed at the tumor-stromal border and assessed semiquantitatively. The absence of staining
reaction in the tumor cells was assessed as a negative result (score 0). Staining in less than 10% of the tumor cells was considered as focal positivy (score 1), and staining in more than 10% of the tumor cells was considered as diffuse positivy (score 2), as ilustrated in figure 1. All patients were followed up until death or every third month for 3 years after surgery and subsequently every sixth month for at least 5 years. Statistical analysis were performed using computer system for biomedical investigation MedCalc (MedCalc Software, Mariakerke, Belgium). A value of p<0,05 was considered statisticaly significant. Student's t-test was
conducted to evaulate differences the means of age. Using the non-parametric Kruskal-Wallis test, expression of MMP-9 was compared to various clinical and histopathological parameters. Survival curves were calculated using Kaplan- Meier method and analysed by log rank test. Univariate Cox regression analysis were performed to determine if the prognostic variables were predictiv of overall survival. Cox analysis were made using BMDP (BMDP Statistical Software Inc., Los Angeles, CA, USA) program package.
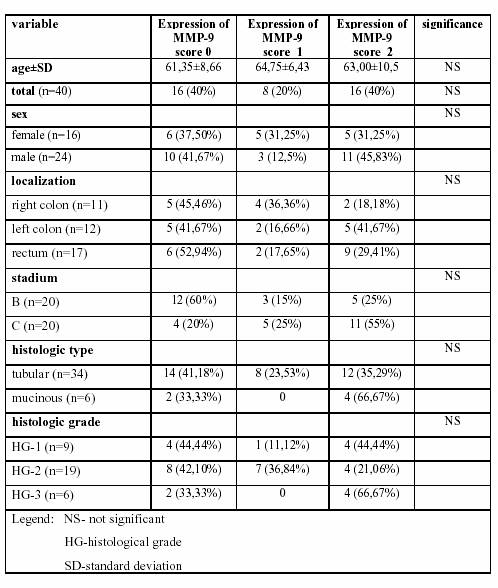
Table
1. Description of clinociopathological data and
associated distribution
of matrix metalloproteinase-9 expression counts
in colorectal cancer.
Table
1. Prognostic influence of clinicopathological
variables by univariate
Cox's proportional hazard regression model for overall
survival in patients with colorectal cancers, with standard
error (SE), 95% confidence interval (CI) and statistical
significance (p). RESULTS The
study was based on 40 patients with CRC out of which
24 were male and 16 were female. The age of the patients
ranged from 33 to 82 years, with mean age of 62,7±8,94
years. Seventeen tumors were located in the rectum, 12
in left colon and 11 in right colon. Histologically among
CRC, there were 9 well differentiated, 19 were moderately,
and 6 were poorly differentiated adenocarcinomas. MMP-9
activity were identified by redish brown intracytoplasmatic
coloration with MMP-9 antibody. The colon
epithelial cells themselves showed negative reaction. Expression
of MMP-9 were not identified in stromal cells (i.e.,
fibroblasts, inflammatory cells, endothelial cells). There
were no differences in staining intensity between the invasive
front of carcinoma compared with central parts. In the
whole study population negative MMP-9 expression in the
colorectal tumor was found in 16 (32%), focal positivity in 8
(16%), and diffuse positivity in 16 (32%) cases. Table
1. shows the major clinicopathologic variables and associated
distribution of MMP-9 expression counts in CRC.
There were no statistically significant associations between
MMP-9 expression and sex, age, localization, stadium of
disease, histological type and histological grade of CRC.
There were increased expressions of MMP-9, but not statistically
significant, in tumor of distant localization (diffuse
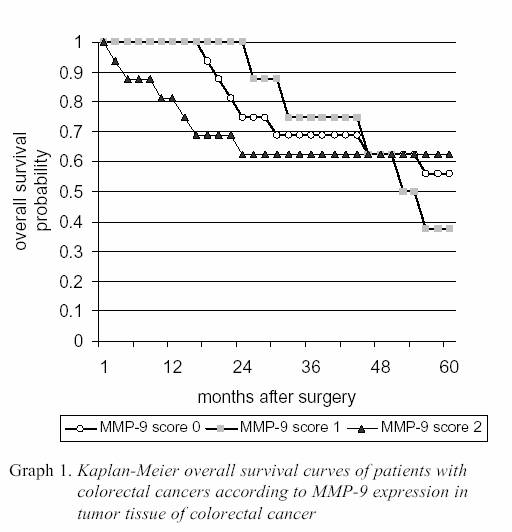
positivity in 2 cases of tumor in right colon, 5 cases in left colon and 9 cases of rectum) and in advanced stage of disease (diffuse positivity in 5 cases of B stadium and 11 cases of C stadium of CRC). Median survival from the time of surgery in whole population was 44.875±19.04 months. The 5 year survival of study population was 55%. The average overall survival in patients with no expression of MMP-9 in CRC was 45.75±17.61 months, in patients with focal positivity of MMP-9 expression 46.62±13.49 months, and in patients with diffuse expression of MMP-9 in tumor tissue 43.12±23.27 months. The 9 (56.25%) patients with negative MMP-9 expression, 3 (37.5%) patients with focal positive MMP-9 expression and 10 (63%) with diffuse MMP-9 expression were alive five years after operation. The Kaplain-Meier survival curves showed no statistically significant differences in the
overall survival rates between patients with different level of MMP-9 expression in CRC tumor cells (Graph 1.). The univariate Cox-models revealed that stage of CRC disease and histologic grade of tumor were prognostic factors for overall survival, while MMP-9 expression in tumor tissue of CRC failed to attain statistical significance (Table 2.).
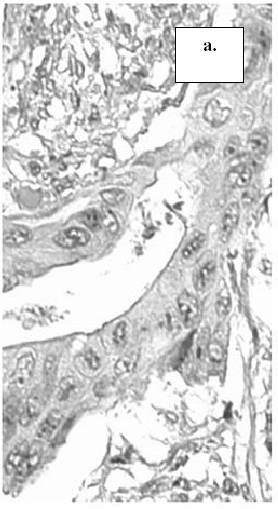
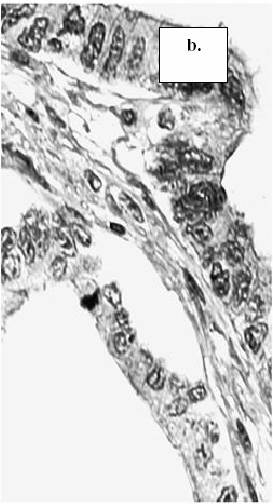
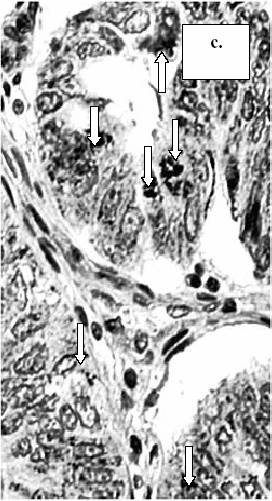
Figure 1. The
expression of MMP-9 in tumor cells of colorectal cancer: a.) absence of staining
reaction in the tumor cells (score 0), b.) staining in less than 10% of the tumor cells
(score 1), c.) staining in more than 10% of the tumor cells (score 2).
(MMP-9, x400) (arrow-head: intracytoplasmatic expression of
MMP-9 in tumor cells). DISCUSSION Metastatic
spread of tumors continues to be a major obstacle
to successful treatment of malignant tumors. Metastasis
is a complex multistep process that requires sequential
interactions between the invasive cell and the ECM
(4). Several studies have been done to try and characterize the
phenotypic and enzymatic profiles of more agressive
tumor cell lines (3). Deteched malignantly transformed cells
migrate and cross structural barriers including BM
and surrounding collagenous ECM. Cellular motility is
associated with controlled proteolysis which involves interaction
between tumor cells and ECM (4). In vivo expression
of MMPs is localized in both tumor and stromal cells
at the invading margin of the tumor, providing a mechanism
for highly concerted degradation of ECM. It may
be that both cellular components contribute to a different part
of the metastatic cascade. The tumor cell MMPs may
contribute to the invasive growth of the tumor while the
stromal element contribute to the remodeling process and
desmoplastic reaction that occur in the tissue adjacent to
the tumor (3). Both gelatinases MMP-2 and MMP-9 are abundantly
expressed in various malignant tumors. MMP- 9 is
mainly expressed by malignant cells, but also by inflammatory
cells, including tissue macrophages and eosinophils
(18). Regulation of the MMPs occurs on three levels:
alteration of gene expression, activation of latent zymogens,
and inhibition by tissue inhibitors of matrix metalloproteinases
(TIMPs). Alteration of all three levels of
control have been associated with tumor cell progression (5).
Most MMPs are secreted as latent precursors (zymogen)
that are proteolytically activated in the extracellulare space.
The activity of MMPs in the extracellular space
is specifically inhibited by TIMPs, which bind to the zinc
binding site of active MMPs at molar equivalence (19).
In malignant disease MMPs are considered to be overexpressed
or TIMPs to be underexpressed, leading to increased
proteolytic activity (3-5). A great deal of evidence has
accumulated in recent years for an important but complex
role of proteases in tumor development. Inhibition
of MMPs provides one attractive target for a novel
class of therapeutic agents to control tumor progression and
metastatic spreading (20). Experimental and preclinical data
are seen to strongly support the ability of MMPs
inhibitors to reduce invasion and spontaneous metastases
and significantly prolonged survival.(21,22) Colorectal
adenocarcinoma is the most frequent cancer in
occidental countries. The mean 5 year relative survival in Europe
remains poor, 40%, in spite of progress in surgery and
chemotherapy (23). Prognosis in patients with CRC conventionally
has been determined by a staging system based
on the extent of primary tumor and the presence or absence
of metastasis, as in Dukes' classification (24). Yet, in
each Dukes' class, the survival of individual patients may vary
considerably. This may occur because the classification reflects
a stage in course of CRC rather than the biologic behavior
of the neoplasma (6). The differences in the survival
rate of patients with the same stage of the CRC disease induced
a search for new diagnostic methods with prognostic
relevance. One of the major field of interest in the
tumor biology is molecular and enzymatic processes in the
invasive margin of tumor. The aim of our study was to assess
the validity of expression of MMP-9 which specifically degrade
the BM as a prognostic marker of CRC. Breakdown
of BM is believed to be an essential step for tumor
invasion and metastases. Despite of many, partly controversial
literature data about intensive proteolytic activity
in CRC in our study we did not find correlation of MMP-9
expression and clinicopathological factor and overall survival
of patients with CRC. Zeng et al. described correlation of
MMP-9 expression with metastatic potential of human
CRC, and he first demonstrated by duoble immunostaining
morphological evidence that type IV collagen degradation
correlates with local elevations in MMP-9 expression
(25,13). In the study of Ambriu et al, patients with
high type IV collagenase activity in CRC tissue had a significantly
shorter disease-free and overall survival time. With
respect to overall survival, only type IV collagenase activity
status provided significant predictive value in multivariate analysis
(26). Garbet et al. reported greater both proteinase
and inhibitor expression in the tumour tissue when
compared with the corresponding normal colorectal tissue
and suggested that increased extracellular proteinase concentrations
and activity may encourage tumour invasion and
metastasis (11). Liabakk et al. found significantly higher expression
of MMP-9 in Dukes' stage A than in stage B carcinoma,
but no correlation with tumor location, differentiation and
disease-free 5-year survival, in contrast to Karakiulakis
et al. who described correlation of MMP-9 activity
to tumor grade, and he also noted enhanced proteolytic activity
in metastases more than in primary tumor (12,14).
Several another members of MMPs family have previously
been investigated in CRC. MMP-2 was frequently studied
in pair with MMP-9 (11-14). Leeman et al. documented
that high MMP-13 staining score showed a trend
towards poorer survival of CRC patients and pointed on
central position of MMP-13 in the MMPs activation cascade, both
activating and being activated by several MMPs (27).
Adachi et al. noted significant correlation of matrilysin
(MMP-7) expression in CRC with the deapth of invasion,
lymph node metastasis, lymphatic invasion, advanced
Dukes' stage and poorer outcome. Matrilysin expression
was a significant prognostic variable for predicting overall
survival in multivariate analysis (28). Shiozawa
et al. investigated MMP-1 immunoreactivity in CRC
found significant correlation with the presence of lymph
node and hepatic metastasis and with increasing stages
of Dukes' classification. The main structural components of
the stroma in the gastrointestinal tract are collagens of
types I and III. Carcinoma cells must break down these structural
components for further invasion through the bowel
wall; such degradation is effected mainly by MMP- 1.
Thus, overexpression of MMP-1 is considered to play a key
role in the process of local tumor invasion (29). In
conclusion, our study did not show significant associations between
intratumoral MMP-9 expression and major
clinicopathological factors and overall survival of patients
with CRC. By univariate Cox hazard-model expression
of MMP-9 did not show prognostic significance. We
think that immunohistochemically staining, that allowes
registration only intracytoplasmatyc, pro forme (92
kDA) of MMP-9 has not shown benefit in pathology analysis
of CRC speciments. Maybe measurement of both pro
and active forme of MMPs, and identification of more than
one MMPs or simultaneously measurement of MMPs and
TIMPs will give better results. |
||
|
|
REFERENCES: 1.
Bray
F, Sankila R, Ferlay J, Parkin DM. Estimates of cancer incidence and
mortality in Europe in 1995. Eur J Cancer 2002; 38:
96-166. 2.
Stetler-Stevenson
WG, Liota LA, Kleiner DE. Extracellular matrix 6: role of matrix
metalloproteinases in tumor invasion and metastasis FASEB J, 1993; 7: 1434-41. 3.
John
A, Tuszynski G. The role of matrix metalloproteinases in tumor angiogenesis and tumor metastasis. Pathology Oncology Research
2001; 7: 14-23. 4.
Böhle
AS, Kalthoff H. Molecular mechanisms of tumor metastasis and angiogenesis.
Langenback's Arch Surg 1999; 384: 133-40. 5.
Westermarck
J, Kahari VM. Regulation of matrix metalloproteinase expression in tumor
invasion. FASEB J 1999; 13: 781-92. 6.
Offerhaus
GJA, Giardiello FM, Brujin JA, Stijnen T, Molyvas EN, Fleuren GJ. The
value of immunohistochemistry for collagen IV
expression in colorectal carcinomas. Cancer 1991;
67: 99- 105. 7.
French
DL, Ramos-Desimone N, Rozinski K, et al. Matrix metalloproteinase-9 in tumor
cell invasion. Ann NY Acad Sci 1994; 732: 324-34. 8.
Chu
YW, Yang PC, Yang SC, et al. Selection
of invasive and metastatic subpopulation
from human lung adenocarcinoma cell line.
Amer J Resp Cell Mol Biol 1997; 17: 353-60. 9.
MacDougall
JR, Bani MR, Lin Y, et al. The 92- kDa gelatinase B is expresed by
advanced stage melanoma cells: supression by somatic
cell hybdridization with early stage melanoma cells. Cancer Res 1995; 55: 4174-81. 10.
Ueda
Y, Imai K, Tsuchiya H, et al. Matrix
metalloproteinase 9 (gelatinase B) is expressed in
multinucleated giant cells of human giant cell tumor
of bone and is associated with vascular
invasion. Am J Pathol 1996; 148: 611-22. |
11.
Garbett
EA, Reed MW, Brown NJ. Proteolysis in colorectal cancer. Mol Pathol 1999; 52:
140-5. 12.
Liabakk
NB, Talbot I, Smith RA, Wilkinson K, Balkwill F. Matrix
metalloproteinase 2 (MMP-2) and matrix metalloproteinase 9 (MMP-9)
type collagenases in colorectal cancer.
Cancer Res 1996; 56: 190-6. 13.
Zeng
ZS, Cohen AM, Guillem JG. Loss of basement membrane type IV collagen is associated with increased expression of
metalloproteinases 2 and 9 (MMP-2 and MMP-9) during human
colorectal tumorigenesis. Carcinogenesis 1999; 20: 749-55. 14.
Karakiulakis
G, Papanikolaou C, Jankovics SM, et al. Increased
type IV collagen-degrading activity in metastases originating from primary
tumors of the human colon. Invasion Metastasis
1997; 17: 158-68. 15.
Sobin
LH, Wittekind Ch, eds. UICC, TNM
classification of malignant tumours. 5th ed. New York: Wiley, 1997. 16.
Astler
VB, Coller FA. The prognostic significance of direct extension of carcinoma of the
colon and rectum. Ann Surg 1954; 139: 846-9. 17.
Bryne
M, Baysen M, Alfsen CG, et al. The
invasive front of carcinomas. The most importan
area for tumor prognosis? Anticancer Res
1998; 18(6B): 4757-64. 18.
Stlhl-Bäckdahl
M, Park WC. 92-kd gelatinase is actively expressed by eosinophils and
stored by neutrophils in squamous cell carcinoma.
Am J Pathol 1993; 142: 995-1000. 19.
Gomez
DE, Alonso DF, Yoshiji H, et al: Tissue inhibitor of metalloproteinases:
structure, regulation and biological function. Europ J Cell
Biol 1997; 74: 111-22. 20.
Massova
I, Kotra LP, Fridman R, Mobashery S. Matrix metalloproteinases: structures,
evolution, and diversification. FASEB J 1998; 12:
1075-95. |
21.
Aparicio
T, Kermorgant S, Dessirier V, Lewin MJM, Lehy T. Matrix
metalloproteinase inhibition prevents colon cancer peritoneal
carcinomatosis development and prolongs survival in
rats. Carcinogenesis 1999; 20: 1445-52. 22.
Rosemurgy
A, Harris J, Langleben A, et al. Marimastat in patients with advanced
pancreatic cancer: a des finding study. Amer J
Clin Oncol 1999; 22: 247-52. 23.
Sant
M, Capocaccia R, Verdecchia A, et al. Comparisons of colon-cancer survival
among European countries: The Eurocare Study.
Int J Cancer 1995; 63: 43-8. 24.
Dukes
CE. The surgical pathology of rectal cancer. J Clin Pathol 1949; 2: 95-8. 25.
Zeng
ZS, Guillem JG. Distinct pattern of matrix metalloproteinase 9 and tissue
inhibitor of metalloproteinase 1 mRNA expression in human colorectal cancer and liver metastases. Br J
Cancer 1995; 72: 575-82. 26.
Ambiru
S, Miyazaki M, Ito H, et al. A
prospective study of prognostic value of type IV
collagenase activity in colorectal cancer tissue.
Dig Dis Sci 1997; 42: 1660-5. 27.
Leeman
MF, McKay JA, Murray GI. Matrix
metalloproteinase 13 activity is associated with poor prognosis in colorectal cancer. J Clinl
Pathol 2002; 55: 758-62. 28.
Adachi
Y, Yamamoto H, Itoh F, Arimura Y, Nishi M, Endo T, Imai K. Clinicopathologic
and prognostic significance of matrilysin expression
at the invasive front in human colorectal
cancers. Int J Cancer 2001; 95: 290-4. 29.
Shiozawa
J, Ito M, Nakayama T, et al. Expression of matrix metalloproteinase-1 in human
colorectal carcinoma. Mod Pathol 2000; 13: 925-33. |