|
|
Alimentary
tract and pancreas Alimentarni
trakt i pankreas ARCH
GASTROENTEROHEPATOL 2003; 22 ( No 1 - 2 ): 5 - 9 |
|||
|
1Njegica
Joji}, 2Jelena
Uroševi}, 1Bo`idar
Boji}, 2Sonja
Pavlovi}. 1Department
of Gastroenterology, Zvezdara
Clinical Center, 2Institute
for Molecular Genetics and Genetic Engineering, Belgrade, Serbia |
. Determination
of thiopurine methyltransferase
genotype in
the patients with inflammatory bowel
disease before and
during azathioprine therapy |
|||
|
Key Words: Thiopurine S-methyltransferase, inflammatory bowel disease, azathioprine, leukopenia. |
Abstract Background:
Azathioprine (AZA) is very effective in treating chronically
active patients with inflammatory
bowel disease (IBD), both Crohn's disease (CD) and ulcerative colitis (UC).
Therapy with
AZA is long lasting and generally safe. Some patients (about 2%) develop
myelosuppresion. Low
activity of enzyme thiopurine S-methyltransverase (TPMT) due to mutation of
TPMT gene is responsible
for bone marrow suppression. Methods:
Polymerase chain reaction - based methods PCR (ARMS) and
RFLP method were used for
determination of TPMT gene mutations. All patients were investigated for 3
most frequent mutant
alleles. The numbers of TPMT gene mutations were analyzed in 35 IBD patients
and control group
consisted of 100 volunteer blood donors. In the IBD group, patients with
myelosuppression were
clinically evaluated. Results:
Thirty-five patients, 24 with CD and 11 with UC enrolled
this study. Twenty-seven were already
on AZA therapy lasting from 1 to 84 mounts; 8 patients were examined before
AZA administration. Thirty-three
( 94.2 % ) cases had normal genotype (TPMT*1/TPMT*1) and 2 ( 5.7 % ) were
heterozygous ( TPMT*1/TPMT*3 ) with one normal ( TPMT*1 ) an one mutant
allele (TPMT*3
). Mutant allele TPMT*3 combined two mutations G460A and A719G. Mutant
alleles TPMT*2,
TPMT*3B and TPMT*3C were not detected. In the IBD group 14 (40 %) patients
had leukopenia
(WBC count <3000/mm3) developed 15 days to 36 mounts after AZA therapy;
one patient
with CD developed serious pancytopenia two weeks after starting AZA. Patient
with early AZA
related pancytopenia had normal genotype. Thirty (85.7%) patients received
concomitant oral 5 -
ASA therapy. In the control group, 4 (4 %) volunteers had mutations; 3 ( 1.5
% ) the most frequent mutant
allele TPMT*3A and 1 (0.5 %) very rare, TPMT*3B. Ninety-six ( 96% )
volunteers had
wild type, normal allele TPMT * 1. Conclusion:
Determination of TPMT activity by genotype could identify
TPMT homozygotes that should
avoid 6MP/AZA in therapy or TPMT heterozygotes that require reduction in
doses. For clinical use
monitoring of WBC counts during therapy was mandatory. Determination of TPMT
genotype should
be reserved for selected cases. |
|||
|
Kljucne
reci: Tiopurine
S- metiltransveraza, inflamatorne
bolesti creva, azatioprin,
leukopenija |
Sazetak Uvod:
Azatioprin
(AZA) je veoma efikasan u le~enju hroni~no aktivnih pacijenata sa
inflamatornim bolestima
creva (IBC), kako Crohn - ove bolesti (CB) tako i ulceroznog kolitisa (UK).
Primena azatioprina
je dugotrajna i bezbedna. Ipak kod oko 2 % pacijenata mo`e da dodje do pojave
mijelosupresije u toku
terapije. Sni`ena aktivnost enzima thiopurine S - metiltransveraze (TPMT )
zbog mutacije
TPMT gena je odgovorna za to. Metod:
Odredjivanje
prisustva mutacije u TPMT genu vr{eno je PCR (ARMS) i RFLP metodom. 35
pacijenata sa IBC i kontrolna grupa koju je ~inilo 100 dobrovoljnih davalaca
krvi testirana su na tri
naj~e{}e mutacije u TPMT genu. Pacijenti kod kojih je do{lo do pojave
lekupenije su posebno klini~ki
evaluirani. Rezultati:
U
studiju je u{lo 35 pacijenata, 24 sa CB i 11 sa UK. 27 pacijenata je bilo na
terapiji AZA u
trajanju od 1 do 84 meseci ; 8 pacijenata je testirano pre zapo~injanja
terapije AZA; 33 ( 94.2%) pacijenta
su imala normalan genotip (TPMT*1/TPMT*1) a 2 (5.7 %) su imala mutirani
genotip (TPMT*1/TPMT/*3A).
Oba su bili heterozigoti sa jednim normalnim TPMT*1 i jednim mutiranim alelom
TPMT*3A. Mutiran alel TPMT*3 sadr`i dve udru`ene mutacije G460A i A719G.
Mutirani aleli
TPMT*2, TPMT*3B i TPMT*3C nisu bili prisutni u grupi pacijenata. 14 ( 40 % )
pacijenata sa IBC je
imalo leukopeniju (WBC < 3,000 mm3) koja se je javila periodu od 15 dana
do 36 meseci nakon
uvodjenja AZA u terapiju. Kod jednog pacijenta sa CB do{lo je do pojave
pancitopenije 15 dana od
primene AZA u dozi od 1mg/kgtt. Ovaj pacijent je imao normalan TPMT genotip.
30 (85.7%)
pacijenata je u terapiji pored AZA dobijalo i neki od oralnih preparata 5
-ASA. U kontrolnoj grupi 96
(96 %) davalaca krvi je imalo normalan TPMT*1 alel. Mutacije su nadjene kod 4
(4%) osobe.
Tri (1,5 %) su imale naj~e{}i mutierani alel TPMT*3A. Jedan (0,5%) davaoc je
imao veoma redak
mutirani alel.TPMT* 3B. Zaklju~ak:
Odredjivanjem
TPMT genotipa mogu da se identifikuju osobe koje su homozigoti za mutaciju
u TPMT genu i kojima ne treba davati u terapiji 6MP/AZA ili heterozigoti kod
kojih se obavezno
redukuje ali ne prekida terapija. Odredjivanje broja leukocita i dalje
predstavlja zlatni standard u
pra}enju pacijenata na terapiji 6-MP/AZA. . |
|||
|
|
INTRODUCTION In
the last few years the use of 6-Mercaptopurine (6MP)
and its prodrug azathioprine (AZA) for the treatment inflammatory
bowel disease (IBD) has been increased.
This therapy is efficacious in the patients with chronic
active Crohn's disease (CD) and ulcerative colitis (UC),
steroid -dependent and steroid refractory, fistulizing CD
and remission maintenance in both diseases (1). It is recognized
that 6-MP and AZA, even at standard dose can cause
severe myelotoxicity (about 1-2 %) in patients with TPMT
(thiopurine S-methyltransverase) deficiency and IBD
(1,2 ). Allelic variants of the TPMT gene are responsible for
the changes in the enzyme activity and possible bone
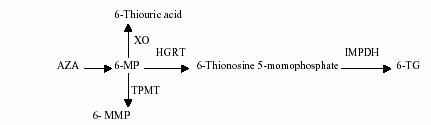
marrow suppression (3,4). AZA and 6-MP are purine analogues.
They have antiproliferative effects on the activated lymphocytes
responsible for inflammation (5). Both drugs
are inactive compounds, which must be converted to 6-thioguanine
(6-TG), active metabolite responsible for immunosuppressive
effect. AZA is absorbed into plasma and
rapidly converted to 6-MP via nonenzymatic pathway 6-MP
is further metabolized by three competitive enzymes:
xanthine oxidase (XO), thiopurine methyltransferase (TPMT)
and hypoxanthine-guanine phosphoribosyltransverase (HGRT).
Only HGRT anabolizes 6-MP into the 6-
thioguanine ( 6-TG ) active nucleotide responsible for the
therapeutic activity and drug related leucopoenia. Reduction
in TPMT activity as a result of allelic variants of TPMT
gene may produce bone marrow suppression because
of preferential metabolism of 6-MP to 6-TG. There
are interindividual and interethnic variations in the TPMT
activity as a result of TPMT gene polymorphism. Most
people ( 88 % ) have genotype with two high (normal) metabolizing
alleles and normally metabolize 6-MP and
AZA. 11 % have one high and one low (mutant) allele and
0.3 % have two low metabolizing alleles (6,7,8). Mutations
occurred alone or in combination on different alleles
of the TPMT gene and may produce functional consequence. We
investigated mutations of TPMT gene in the IBD
patients who were on AZA therapy and its practical and
clinically use in the monitoring myelosuppression. MATERIAL
AND METHODS From
January 2002 to April 2003, 35 patients with proven
IBD were analyzed, 24 with CD and 11 with UC. Twenty-seven
were already on oral AZA therapy lasting from
1 to 84 months. Eight patients were examined before AZA
administration. All patients had complete blood counts
measured weekly for the first month and then monthly
for the next months. TPMT gene mutations were analyzed
in the IBD patients and control group consisted of 100
volunteer blood donors. Polymerase chain reaction - based
method ( PCR ) Amplification Refractory Mutation System
( ARMS ) and Restriction Fragment Length Polymorphism
( RFLP ) method were used for determination of
TPMT gene mutations (6,7). This strategy allows identifying
known mutations of the gene. DNA sample were
investigated for normal and three most frequent mutant
alleles. Point mutations of the gene were known to occur
either on the same allele or on the different alleles. The
most frequent mutant allele in white population TPMT*3A
combined two mutations G460A and A719G on the
same allele (6, 8, 9). Identification of genotype can predict TPMT
phenotype and recognize the patients at higher risk
of myelotoxicity during AZA/6-MP therapy (6). Some of
these TPMT alleles are functional as *1, *1S, and *1A, or
nonfunctional as *2, *3A, *3B, *3C, *3D, *4, *5, *6, *7.
Individuals may be homozygous or heterozygous for one
or two functional or nonfunctional mutations, which predict
their TPMT genotype associated with enzyme activity.
For statistical analysis descriptive statistics, frequencies and
group analysis were performed.
Figure
1. Azathioprine ( AZA ) metabolism. RESULTS Between
January 2002 and April 2003 there were 35 patients
with IBD who had TPMT genotyping ( 24 with CD
and 11 with UC, 22 females, 13 males, ages 19 - 48, median
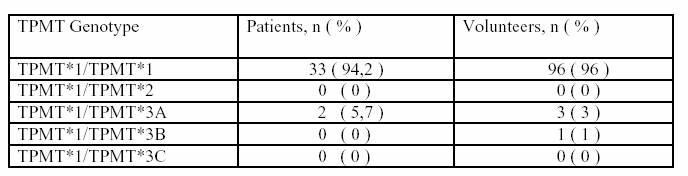
31 ). Thirty-three ( 94,2 % ) patients were homozygous with
two wild type ( normal ) TPMT*1/TPMT*1 alleles corresponding
to high enzyme activity. Two ( 5,7 % ) patients,
one with CD and one with UC were heterozygous (
TPMT*1/TPMT*3 ) with one wild type TPMT*1 and one mutant
allele TPMT*3A ( Table 1 ). This mutant allele combines
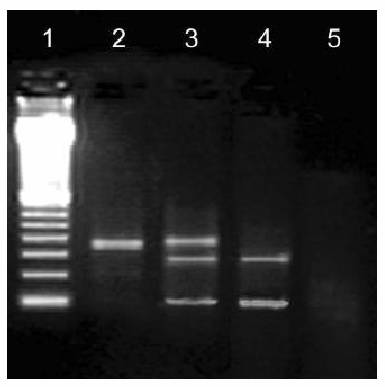
the two mutations: G460A, which results guanine to
adenine substitutions at codon 460 ( G460 ? A ), and
A719G adenine to guanine substitution at codon 719 ( A719
G ) ( Figure 2 ). These mutations correspond to intermediate
enzyme activity. Genotype TPMT*1/TPMT*2,
TPMT*1/TPMT*3B and TPMT*1/TPMT*3C
were not detected in IBD group. Frequency
of TPMT*1 normal allele were 97.1 % and of TPMT*3
mutant allele 2.9 % . We also determined TPMT genotype
in the control group consisting of 100 volunteer blood
donors. 96 ( 96 % ) volunteer had normal genotype TPMT
*1/TPMT *1. Four ( 4 % ) had mutant genotypes, 3 (
3 % ) TPMT *1/TPMT *3 and 1 (1 % ) very rare TPMT*1/TPMT*3B
genotype. Frequency of TPMT* 1 normal
allele in the control group were 98%, mutant alleles TPMT*3
1.5 % and TPMT *3B 0.5 %. Twenty-seven IBD
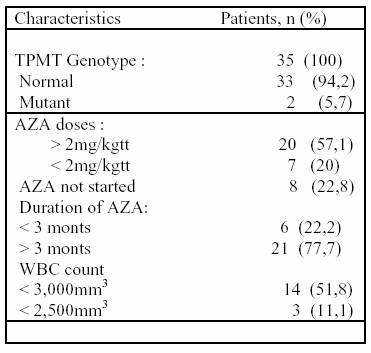
patients received AZA therapy in dose of 1 - 2.5mg/kgtt
lasting from 1 to 84 mounts. Eight patients were
examining before AZA therapy. In the IBD group 14 patients
had leukopenia ( WBC < 3,000/mm3 ) developed 15
days to 36 mounts after starting AZA. Three patients had
serious leukopenia ( WBC < 2,500/ mm3 ) receiving doses
of AZA between 1.5 - 2mg/kgtt, two, eight, nine and
16 mounts after starting therapy ( Table 2 ). Thirty patients
received concomitant 5- ASA therapy in dose of 1.5
- 4 g/day. Only one patient with CD had pancitopenia two
weeks after AZA therapy at dose of 1mg/kgtt. This patient
stopped AZA immediately. She didn't have any concomitant
medication during AZA therapy and her TPMT
genotype was normal ( TPMT*1/TPMT*1 ). She was
on Sulphasalazine ( SASP) 3g/day one year ago and stop
with them three mont before starting AZA During SASP
therapy leukopenia was not developing. Her hematologist confirms
normal cellularity of bone marrow. Patients
with mutant genotype ( TPMT*1/TPMT*3 ) had WBC
counts between 2.600 - 3.000/ mm3 during AZA therapy.
Patient with CD and mutant genotype received AZA
in dose of 125mg/day for 11 monts. Patient with UC and
mutant genotype was on the same AZA dose for 24 mounts.
Both patients had concomitant medication, one with
5-ASA in dose of 1,5g/day and second one with SASP in
dose of 3g/day. Daily AZA dose was reduced in both patients
but not stopped. 5-ASA and SASP therapy was started
three and five years ago but patients never developed leukopenia.
None of the 8 patients examining before starting
AZA therapy developed TPMT gene mutation.
Figure
2. Detection of G460A mutation in TPMT gene with
PCR RFLP1 method.
Table
1. Distribution of TPMT genotype in IBD patients and
control group.
Table
1. AZA dosage and WBC count in IBD patients with
normal and mutant TPMT genotype DISCUSSION In
the last few years 6MP/AZA is widely used for the treatment
of chronic active IBD, mostly CD. This therapy is
long lasting and safe. About 2 % of patients with TPMT enzyme
deficiency are intolerant to AZA and may develop leukopenia
during the treatment (1.2). Bone marrow suppression is
due to excessive accumulation of cytotoxic metabolites
as a result of higher 6-MP conversion to 6- thioguanine
nucleotides (10,11). These patients should not receive
6-MP or AZA for IBD treatment. This was the reason for
TPMT analysis in IBD patients (12). Colombel et al.
confirmed that myelosuppression occurred within 1.5 months
in TPMT homozygous patients, but much more later
in heterozygous or in patients without mutations (3). TPMT
enzyme activity may be determined by genotype or phenotype
which means by direct measurement of red blood
cell (RBC) enzyme activity . In the beginning genotyping was
the only commercially available test. Today determination
of TPMT phenotype is also commercially available
and may provide a more accurate assessment of enzyme
activity (4). To make a choice, one or the other method
depends on laboratory possibilities. We investigated TPMT
genotyping in 35 IBD patients during AZA therapy and
found that only 2 (5.7%) patients were heterozygous with
one wild (TPMT*1) and one mutant (TPMT*3) allele
that correspond to intermediate TPMT activity (3,4). In
the control group 4 (4%) individuals had TPMT gene mutations.
Frequency is similar as in a European control population
where 10 % had mutations (12). Fourteen IBD patients
during AZA therapy developed leukopenia (WBC <
3.000 mm3), only two patients had TPMT gene mutations. They
were 11 and 24 mounts on AZA (2mg/kgtt), without
serious leukopenia during monitoring. Both patients
were heterozygous with nonfunctional TPMT*3A alleles
and intermediate enzyme activity. This was the reason to
reduce daily AZA dose.Mutations that correspond to alleles
*2, *3A and *3C are most frequent in Europe (12). All
patients in our study with leukopenia also had concomitant oral
5 -ASA therapy that has been shown to inhibit
TPMT activity and increase the serum 6-TG levels and
predispose patients with AZA therapy to leukopenia overlooked
etiology was possible in the majority of these patients. In
conclusion: determination of TPMT activity by genotype
could identify TPMT homozygotes that should avoid
6MP/AZA in therapy or TPMT heterozygotes that require
reduction in doses and careful introduction of the other
drugs that may interfere with myelotoxicity during AZA
therapy. For clinical use monitoring of the WBC and platelet
counts during 6MP/AZA therapy was mandatory. Determination
of genotype or phenotype is much more expensive
and should be reserved for selected patients only. (13,14).
Only one patient developed serious pancytopenia 15
days after AZA administration at dose of 1mg/kgtt. This
patient had normal TPMT genotype. She was on SASP
for two years and stopped with them three mounts before
being treated with AZA . She also received metronidazol eight
monts before AZA therapy. Both drugs may cause
myelosuppression (13,15). Her hematologist excluded
bone marrow irregularity. She did not have any viral
infection known to induce neutropenia or trombopenia (
16,17). TPMT phenotype or direct measurement of enzyme
activity may provide a more accurate assessment of
metabolic activity and toxicity in these patients (18). Finally,
no obvious cause of bone marrow suppression or |
|||
|
|
REFERENCES: 1.
Sanborn
WJ. Azathioprine : State of the art in inflammatory bowel disease. Scand J Gastroenterol Suppl 1998; 225 : 92-9. 2.
Cuffari
C . Azathioprine metabolism in inflammatory bowel disease. Proceedings of the Falk Symposium 111, Frieburg 2000; 226-235. 3.
Colombel
JF, Ferrari N, Debuysere H, Marteau P. gendre JP, Bonaz B, Soule JC,
Modigliani r, Touze Y, Catala P, Libersa C, Broly
F. Genotypic analysis of thiopurine S- methyltransverase in
patients with Crohn's disease and severe
myelosuppression during azathioprine therapy. Gastroenterology 2000; 118(6):1025-30. 4.
Regueiro
M, Mardini H. determination of Thiopurine Methyltransverase Genotype
or Phenotype Optimizes Initial Dosing of Azathioprine for the Treatment of
Crohn's Disease. 5.
Sanborn
W, Sutherland L, Person D, et al. Azathioprine os 6-mercaptopurine for
inducing remission of Crohn's disease. The
Cohrane Database of Systemic Reviews Issue 2,
2002. 6.
Yates
CR, Krynetski EY, Loennechen T, fessing MY, Tai HL, Piu CH, Relling
MV, Evans WE. Molecular diagnosis of thiopurine
S-methyltransverase deficiency:genetic basis for
azathioprine and mercaptopurine intolerance. Ann
Intern med 1997; 126; 608-614. |
7.
Tai
Hl, Krynetski EY, Yates CR, Loennechen T, Fessing MY, Krynetskaia NF,
Evans WE. Thiopurine S - methyltransverase
deficiency : two nucleotid transitions define the most
prevalent mutant allele associated with loss of
catalytic activity in Caucasians. Am J Hum Genet
1996; 58 : 694-702. 8.
Otternes
D, Szumlanski C, Lennard L, Klemetsdal B, arbakke J, Park-Hah JO,
Iven H, Schmiegelow K, Branum E, O'Brien J,
Weinshilboum R. Human thiopurine methyltransverase
pharmacogenetics : gene sequence polymorphism. Clin
Pharmacol ther 1997;62:60-73. 9.
Spire
Vayron de la Moureyre C, Debuysere H, Mastain B, Vinner E, Marez D,
Lo Guidice JM, Chevalier D, Brique S, Motte
K, Colombel JF, Turck D, Noel C, Flipo RM,
Pol A, Lhermitte M, Lafitte JJ, Libersa C, Broly
F. Genotypic and phenotypic analysis of the thiopurine
S-methyltransverase gene (TPMT) in a European population.
Br J Pharmacol 1998; 125;879-887. 10.
Kaskas
BA, Louis E, Hindorf U, Schaeffeler E, deflander J, Greapler F,
Schmiegelov K, Gregor M, Zanger UM, Eichelbaum M,
Schwab M. Safe treatment of thiopurine S-
methyltransferase deficient Crohn's disease patients with azathioprine. Gut 2003; 52: 140-142. 11.
Lennard
L, Lilleyman JS, Van Loon J, Weinshilboum RM. Gernetic variation in response to 6-mercaptopurine for childhood acute
lymphoblastic leukaemia. Lancet 1990;336:225-229. |
12.
Szumlanski
C, Otterness D, Her C, Lee D, Brandriff B, Kelsell D, Spurr
N, Lennard L, Weiben E, Weinshilboum RM. Thiopurine methyltransverase pharmacogenetics: human gene cloning and characterization of a
common polymoprhism. DNA Cell Biol 1996; 15: 17-30. 13.
Szulmanski
CL, Weinshilboum RM. Sulphasalazine inhibition of thiopurine
methyltransverase: possible mechanisam for interaction with 6 - mercaptopurine and
azathioprine. Br J Clin Pharmacol 1995;39:456-9. 14.
Lewis
LD, Benin A, Szumlanski CL at al. Olsalazine and 6-mercaptopurine related
bone marrow suppression; a possible
drug-drug interaction. Clin Pharmacol Ther 1997;62:464-75. 15.
Smith
JA. Neutropenia associated with
metronidazole therapy (latter). CMAJ 1980;123-202. 16.
Feusner
JH, Slicher SJ, Harker LA. Mechanisms of tromobocytopenia in varicella. Am J
Hematol 1979;7:255-264. 17.
Veraldi
S, Rizziteli G, Lunghi G, Gardone R. Primary infection by human parvovirus
B19. Dermatology 1993;186:72-74. 18.
Seidman
EG. Clinical use and practical application of TPMT enzyme an mercaptopurine
metabolite monitoring in IBC. Rev. Gastroenterol
Disord 2003;3 Supll 1:S30-8. |
|