Alimentary tract and pancreas
Alimentarni trakt i pankreas
ARCH GASTROENTEROHEPATOLOGY 2000; 19 ( No 3 – 4 ):
CLINICAL USEFULNESS OF ENDOSCOPIC ULTRASONOGRAPHY IN DIFFERENTIAL DIAGNOSIS OF SUBMUCOSAL OESOPHAGO-GASTRIC TUMOURS
Klinicki znacaj endoskopske ultrasonografije u dijagnostici submukoznih tumora jednjaka i zeludca
Endoscopic ultrasonography and GI submucosal tumours
( accepted November 26th, 2000 )
1Miodrag Krstic, 2Petar Pesko, 1Predrag Dugalic, 1Dragan Tomic, 1Tomica Milosavljevic, 3Slobodan Krstic, 1 Aleksdandra Pavlovic, 1Milan Spuran, 1Marjan Micev ,1Mirko Bulajic.
1Institute of Digestive Diseases, Clinic of Gastroenterology and Hepatology, 2 Center for Oesophageal Surgery, Institute of Digestive Diseases, Clinical Center of Serbia, 3 Emergency Center, Clinical Center of Serbia, Belgrade.
Address correspondence to: Docent Dr Miodrag Krstic
Institute of Digestive Diseases
6 Koste Todorovica St.
YU-11000 Belgrade, Serbia,
Yugoslavia
E-mail:miodrag [email protected]
Abbreviations used in this article: EUS, endoscopic ultrasonography; SML, submucosal lesion; GIST, GI stromal tumours.
ABSTRACT
In order to determine the usefulness of endoscopic ultrasonography (EUS) in differential
diagnosis of upper GI submucosal tumours EUS was performed in 350 consecutive
patients. The location, size, echo pattern, originating layer of submucosal lesion and regularity of its outer margin were explored. The results were compared with CT, angiography, operative findings, and tumour histology.
Twenty-nine stromal tumours originating from muscularis propria of the fundus ( 9 ), stomach body ( 9 ), oesophagus ( 8 ), postbulbar duodenum ( 2 ), and papilla of Vater ( 1 ) were diagnosed. In a single case tumour originated from lamina muscularis mucosae was diagnosed as well. Fourteen patients were operated and in 9 cases EUS diagnosis of benign stromal tumor was confirmed. In 5 patients EUS correctly revealed malignant fundic ( 3 ) and corporal ( 2 ) submucosal gastric tumours.
Other EUS diagnoses were as follow: oesophageal lipoma ( 1 ), submucosal metastases of malignant melanoma ( 2 ), duodenal bulb carcinoid ( 1 ), abberant pancreas ( 1 ), extraluminal compression mimicking submucosal tumour ( 2 ), antral submucosal cysts ( 2 ), fundic varices ( 4 ). In a single patient, pancreatic psudocyst was incorrectly diagnosed as a submucosal cyst of the stomach body.
Key words: endoscopic ultrasonography, submucosal GI tumours.
SAZETAK
U cilju procene korisnosti endoskopske ultrasonografije u diferencijalnoj dijagnozi submukozalnih tumora proksimalnih delova GI trakta, ovi pregledi su nacinjeni u 350 pacijenata. Mesto, velicina, ultrasonografske osobine, ishodisna struktura submukoznih promena, i pravilnost spoljne ivice utvrdjenih promena je ispitana. Rezultati su uporedjeni sa CT nalazom, angiografijama, operativnim nalazom, i histologijom tumora.
Dvadest i dva stromalna tumora porekla misicnog sloja fundusa ( 9 ), tela zeludca ( 9 ), jednjaka ( 8 ), postbulbarnog duodenuma ( 2 ), i papile Vateri ( 1 ) su dijagnostikovani. U jednom slucaju tumor je bio porekla lamine muscularis mukoze. Cetrnaestoro pacijenata je bilo operisano. U 9 endoskopska ultrasonografska dijagnoza benignog stromalnog tumora je potvrdjena. U 5 slucajeva ovom metodom je korektrno otkriven maligni fundusni ( 3 ) odnosno submukozni tumor tela zeludca ( 2 ).
Ostale endoskopske ultrasonogfarske dijagnoze su bile: lipom jednjaka ( 1 ), submukozne metastaze malignog melanoma ( 2 ), karcinoid bulbusa duodenuma ( 1 ), aberantni pankreas ( 1 ), ekstraluminalna kompresija ( 2 ), antralna submukozna cista ( 2 ), fundusni variksi ( 4 ). U 1 slucaju nalaz pankreasne pseudociste je neodgovajuce protumacen kao submukozna cista zida tela zeludca.
Kljucne reci: endfoskopska ultrasonografija, submukozni GI tumori.
INTRODUCTION
Routine diagnosis of GI mucosal abnormalities are based on conventional endoscopy and mucosal histology ( 1 ). However, GI submucosal lesions ( SML ) are oftenly diagnostic and therapeutic problem because standard endoscopic tissue specimens are usually superficial for adequate microscopic analysis ( 2 ). Endoscopic ultrasonography ( EUS ) allows high-resolution, cross-sectional demonstration of the entire gut wall ( 3 ) Since its introduction, the diagnosis of SML become more accurate ( 4-7 ). EUS allows clear distinction between intramural lesions and extramural compression ( 4 ) It also provides measurement of the real size of the SML, location of originating layer, reggularity of outer margin, and finaly presumption of its histological nature ( 5-7 ). Here we present our analysis of clinical usefulness of the EUS in differential diagnosis of oesophageasl and gastric submucosal tumours..PATIENTS AND METHODS
From September 1998 to September 2000, EUS was performed in 350 consecutive patients. In 44 the indication for EUS was clinical suspicion to SML by conventional endosopy. All EUS examinations were performed using Olympus GIF-130 videoecho-endoscope with 7,5/12MHz switchable radial probe. For each case the location, size, originating layer, echo pattern and regularity of the outer margins of suspected SML were recorded. The results were compared with CT, angiography and operation with histology when possible.
Results
Gastrointestinal stromal tumours ( GIST ) originating from 4th ( muscularis propria ) or 2nd ( muscularis mucose ) layers were diagnosed in 30 patients by EUS.. Table 1 and 2. Figure 1 and 2. In 29 patients, GIST arrose from muscularis propria and overlying mucosal and submucosal layers were identified by EUS. Figure 1 . In a single patient, GIST originated from muscularis mucose. Nine GIST were located in distal oesophagus, 18 in the stomach, and 3 in the duodenum. Table 2. No GIST was identified in the stomach antrum. In all cases tumour length varied from 12-57mm, depth 8-35mm.
EUS features of benign GIST, e.g.homogenicity, hypoechoic structure, regular outer margin, and diametar less than 40mm, fullfilled SML in 25 patients. Table 1 and 2. Nine patients with EUS diagnosis of benign GIST were operated because of reccurent GI bleeding and/or obstruction. EUS diagnosis was confirmed in all cases by histology and immunochistochemistry.. All symptomatic GIST were larger than 25mm in diametar. In 16 symptom-free patients, CT revealed the existance of probable submucosal tumour. These patients are on regular EUS checks-up every 6 months.
EUS features suggesting malignancy, e.g. diametar larger than 40mm, irregular outer margin, inhomogenicity with cystic spaces inside the tumor, were diagnosed in 5 patients. Figure 2, 3,4. All were opreated and tumour histology confirmed EUS diagnosis in all cases.
In one patient oesophageal lipoma was diagnosed by EUS. Figure 5. EUS features of lipoma were: peristentence of hyperechoic tumour in submucosa. The patient is on regular follow-up.
In 3 patients, EUS diagnosis of the antral submucosal cyst was made. This was confirmed on operation in 2 cases. In a single patient, large, penetrating, pancreatic pseudocyst was incorrectly diagnosed as gastric submucosal cyst. In 4 patients, EUS showed varices in the fundus of the stomach as multiple, small, anechoic, round structures arrising in the submucosa. Figure 6a,b. Such finding was confirmed on angiography in all cases.
In 2 patients, extraluminal compression mimicking SML was correctly diagnosed by EUS. In this cases EUS finding of intact gut wall with five uninterrupted layers ruled out the SML. This was confirmed by abdominal CT which showed enlarged liver compressing the stomach wall.
In 2 patients, submucosal metastases of malignant melanoma were visualized as submucosal hypoechogenic oval lesions. Figure 7. EUS diagnosis was confirmed by endoscopic biopsy. In a single patient, malignant carcinoid tumor of duodenal bulb was clearly demonstrated as well defined submucosal lesion of intermediate echogenicity. Figure 10. Finaly, in one patient, the abberant pancreas presented as an antral submucosal tumor of mixed structure in submucosa and muscularis propria.
Discussion
GIST are the most common alimentary submucosal tumours ( 8 ). Two thirds are gastric, mostly in the upper part of the stomach ( 9 ). According to some autopsy series their incidence is 50% in general population ( 10 ) However, 90% are lesser than 5mm and clinically silent. Progressively smaller number of GIST are of small intestinal, colonic, and oesophageal origin ( 11,12 ). In this study we confirmed literature data that GIST are the most common SML ( 30 – 41 ). We further comfirmed that the majority gastric GISTS are located in the upper part of the stromach. In our series, oesophageal GIST were also single and located in the supracardial region.
EUS typically shows GIST to be hypoechogenic mass originating from muscularis propria ( 4th layer ) or rarely from muscularis mucosa ( 2nd layer ) ( 1,9,12 ). In our series originating layer was clearly delineated by EUS in all patients: in 29 this was muscularis propria and in 1 muscularis mucosa. EUS features associated with benign GIST are: size less than 40mm, regular outer margin, and homogenicity of the echo pattern ( 8,9,12 ). Such criteria fulfilled 25 patients. Nine of them with symptoms of GI bleeding and/or obstruction were operated. EUS diagnoses was confirmed histologically in all of them. In this series symptomatic benign GIST were larger than 25mm in diameter. Five patients showed EUS signs suggestive for malignancy: >40mm, irregular outer margin, and inhomogenicity with cystic spaces inside the tumour ( 1,9,12 ) This was confirmed at operation, which disclosed the malignant GIST.
However, neither EUS, nor EUS-FNA cannot accurately predict malignant versus benign behaviourof GIST with full certainity ( 11,12 ). Therefore, symtomatic benign and malignant GIST have to be operated. Rational menagement of small, asymptomatic GIST lesser than 30mm is still contraversial ( 9 - 12 ). This lesions might never became clinically significant, especially given their high incidence at autopsy ( 10 ). On the other hand, caution must be given to any GIST because there are reports of metastatic disease occuring years after surgical removal of small, benign appearing tumors ( 12 ). Thus it looks prudent that serial EUS follow-up is an alternative to surgery. Continuing growth and possible malignant transformation require prompt surgery. This approach we currently employed in our group of symptom-free patients.
The term GIST encompases a heterogenous group of mesenchimal GI neoplasms previously referred as schwannomas, leiomyomas, and leiomyosarcomas ( 13 ). They can arrise anywhere along the GI tract, from the oesophagus to the rectum ( 14 ). They were originally considered to be of smooth muscle origin but further studies showed little evidence of smooth muscle or neural defferentiation ( 15 - 22 ). Although heterogenous in morphology, GIST represent a family of tumours probably originating from the intestinal pace-maker cell, interstitial cell of Cajal ( 15 ). Differential diagnosis of these tumours include smooth muscle tumours, schwannomas, and pure GIST. The first two are exceedingly rare. It appears that most of reported cases of gastric leiomyomas and schwannomas probably belong to pure GIST category ( 16 – 23 ). The tumour cell phenotype can be established only when histomorphology is coupled with immunostaining and sole demonstration of smooth muscle on histology is not sufficient to make a diagnosis of smooth muscle tumor ( 17 ). Predicting the malignancy is very difficult due to slow-growing nature of all GISTs ( 18 ). The prognosis in individual cases of GIST is most commonly the result of a rather complex interrelation between patient’s age, tumor size and location, EUS finding, mitotic index, DNA ploidy, cytogenetics and immunophenotyping ( 13,19 ).
Oesophageal lipomas constitute less than 5% of benign oesophageal tumours ( 11 ). They are benign growth of mature lipocytes, without any cellular atypia and are located in the submucosa. EUS shows a characteristic hyperechoic mass located in 3rd layer ( submucosa ) ( 1,9,12 ). This was confirmed in one our patient. Malignant potential of these lesions is extremely low or nonexistent.
Gastric submucosal cysts are rare. They are sharply demarcated echo-free lesions with dorsal acoustic accentuation sandwiched between the two hypoechoic wall layers ( muscularis propria and muscularis mucose ) ( 1,12,19 ). We correctly visualized two cysts by EUS. In a single patients large pancreatic pseudocyst penetrating the entire gastric wall mimicked submucosal cyst.
Fundic varices are localized in submucosa, too ( 3rd layer ) ( 9 ). Since fundic mucosa is usually thickened, endoscopic visualisation might be very difficult. It was already reported that EUS could detect fundic varices earlier and more oftenly than conventional endoscopy ( 24-25 ). On EUS, they appear as small, round to oval, echo-free structures. We diagnosed 4 patients with isolated gastric varices due to left-sided portal hypertension. EUS finding was confirmed on angiography.
Endoscopic ultrasonography can readily distinguish gastric wall impressions from submucosal tumours ( 9 ). In our 2 patients EUS correctly recognised enlarged viscera as a cause of suspected SML.
Metastatic submucosal tumours and carcinoids are rare. They present as oval, echo-poor, relatively demarcated foci in 3rd layer ( submucosa ) ( 26 ). In the most cases muscularis propria remains intact as it happened in our three patients.
EUS is currently the most reliable technique for detection of submucosal tumors and their differentiation from extraluminal processes. It can provide diagnostic information about the type of submucosal lesion ( tumour, cyst, varices ) and sometimes predict its histological nature ( GIST, lipoma, carcinoid ). Our data further confirmed the importance of EUS in management of patients with suspected SML.
REFERENCES:
Caletti G, Fusaroli P, Bocus P. Endoscopic Ultrasonography. Digestion 1998; 59: 509-530.
Chak A. Endoscopic Ultrasonography. Endoscopy 2000; 32: 146-152.
Darles Y, Clouard R. Physical principles: endosonographic apperance of the normal gastrointestinal tract wall. In: Dancygier H, Lightdale J. Endosonography in gastroenterology Ied. Stuttgart-New York: Thieme-Verlag; 1999:1-12.
Yasuda K, Nakajima M,Yoshida S et al. The diagnosis of submucosal tumors of the stomach by endoscopic ultrasonography. Gastrointest.Endosc 1989; 35:10-15.
Yasuda K,Cho E, Nakajima m et al. The diagnosis of submucosal lessions of the upper gastrointestinal tract by endoscopic ultrasonography. Gastrointest Endoscopy 1990; 36: S17-20
Caletti G, Zani I, Bolondi L, et al. Endoscopic ultrasonography in the diagnosis of gastric submucosal tumors. Gastrointest Endosc 1989;35:413-418
Elisabetta Buscarini, MD, Michele Di Stasi, MD, Sandro Rossi, MD, Matteo Silva, MD, Francesco Giangregorio, MD, Zangrandi Adriano, MD, Luigi Buscarini, MD Endosonographic diagnosis of submucosal upper gastrointestinal tract lesions and large fold gastropathies by catheter ultrasound probe Gastrointest Endosc 1999;49:184-91.
Yasuda K. Submucosal tumors of the upper gastrointestinal tract:Evaluation by the endoscopic ultrasonography. In: Morton A, et al. Neoplasms of the digestive tract:Imaging, staging and menagement Ied. Raven: Lippincott;1998:85-92.
Dancygier H, Lightdale C, Stevens P.Endoscopic ultrasonography of the upper gastrointestinal tract and colon. In: Dancygier H, Lightdale J. Endosonography in gastroenterology Ied. Stuttgart-New York: Thieme-Verlag; 1999: 13-174.
Yasuda K, Nakajima M, Kawai K. Endoscopic ultrasonographic imaging of submucosal lessions of the upper gastrointestinal tract. Gastrointest Endoscopic Clin North am 1992;2: 615-24.
Boland R. Tumors of the stomach. In:Yamada T et al. Textbook and atlas of gastroenterology. IInd ed. New York. Lipnicott Wiliams and Wilkins. 1999:227-36.
Yasuda K. Gastrointestinal lesions. In:Yasuda K. The handbook of endoscopic ultrasonography in digestive tract. Ist Ed Tokio:Blackwell Science Japan;2000:45-63.
Boggino H, Fernandez M and Logrono H. Cytomorphology of Gastrointestinal stromal tumor. Diagnostic cytomorphology 2000; 23: 156-160.
Sarlomo-Rikala M, Kovatich AJ, Miettinen M, et al. CD117: a sensitive marker for gastrointestinal stromal tumors that is more specific than CD34. Mod Pathol 1998;11:728-34.
Sircar K, Hewlett BR,Huizinga JD, Chorneyco K, et al. Interstitial cells of Cajal as precursors of gastrointestinal stromal tumors. Am J Surg Pathol 1999;23:377-389.
Cunningham RE, Federspiel BH, McCarthy WF et al. Predicting prognosis of gastrointestinal smooth muscle tumors; role of clinical and histologic evaluation, flow cytometry, and image cytometry. Am J Surg pathol 1993; 17:588-594.
Emory TS, Sobin LH, Lukes L, et al. Prognosis of gastrointestinal smooth-muscle(stromal) tumors: dependence of antomic site. Am J Surg pathol 1999;23:82-87.
El-Rifai W, Sarlomo-Rikala M, Miettinen M, et al. DNA copy number losses in chromosome 14: an early change in gastrointestinal stromal tumors. Cancer Res 1996;56:3230-3232.
Tao LC, Davidson DD. Aspiration biopsy cytology of smooth muscle tumors: a cytologic approach to the differentiation between leiomyosarcoma and leiomyoma. Acta Cyol 1993;37:300-308.
Das DK, Pant CS. Fine needle aspiration cytologic diagnosis of gastrointestinal tract lesions: a study of 78 cases. Acta Cytol 1994; 38:723-729.
Miettinen M, Virolaoinen M, Sarlomo-Rikala M. Gastrointestinal stromal tumors-value of CD34 antigen in their identification and separation from true leimyomas and scwannomas. Am J Surg Pathol 1995;19:207-216.
Huliramn J, Gradual D. Gastrointestinal stromal tumors: an immunohistochemical study of 165 cases. Histopathology 1991;19:311-320.
Prevot S, Bievenu L, Vaillant JC, de Saint-Maur PP. Benign schwannomas of the digestive tract: a clinipathologic and immunohistochemical study of five cases, including a case of esopphaegal tumor. Am J Surg Pathol 1999;23: 431-436.
Caletti C, Brocchi M, Balardini M, et al. Assessment of portal hypertension by endoscopic ultrasonography. Endoscopy 1990; 36:21-27
Nattermann Ch, Dancyiger C. Endoskopishe Sonographie bei portaler Hypertension. Endosk.Heute 5 1992:26-29.
Yoshikane H,Tsukamoto Y, Niwa Y et al. Carcinoid tumors of the gastrointestinal tract: evaluation with endoscopic ultrasonography. Gastrointest.Endosc 1993;39: 375-383.
Table 1. The site of origin and EUS appearance of various
submucosal lesions.
| Type of lesion | No. | EUS appearances |
| Benign GIST Malignant GIST Lipoma Cyst Varices Abberant pancreas Metastases of melanoma Carcinoid | 25 5 1 2 4 1 2 1 | Hypoechoic,homogenous, originate from 4th (m.propria) or 2nd (m.mucosa) layer, >4cm;irregular outer margin, cystic spaces, originate from 4th or 2nd layer Hyperechoic; originate in 3rd (submucosa) layer Round, well defined, anechoic, echo-free internal structure, located in 3rd (submucosa) layer Multiple, anechoic or hypoechoic, located in submucosa Irregular echo pattern; mixt structure, located in submucosa Hypoechoic, located in submucosa or m.propria Intermediate echogenicity, located in submucosa |
__________________________________________________________
Table 2. Location of SML in our group patients
| Type of lesion | Oesophagus _________stomach duodenum _________ Fundus body antrum |
| Benign GIST Malignant GIST Lipoma Cyst Varices Abberant pancreas Metastases of melanoma Carcinoid | 9 7 3 2 1 2 4 1 2
1 |
________________________________________________________
FIGURES:
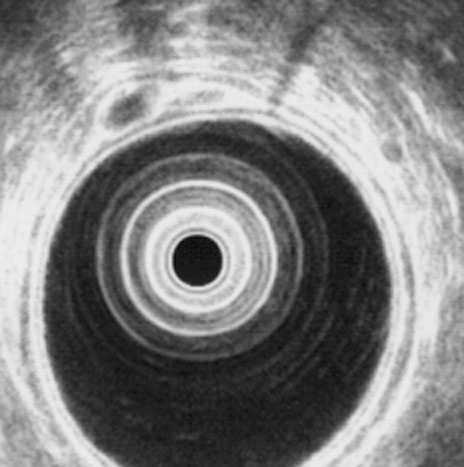
Figure 1. Small benign GIST of the stomach body.
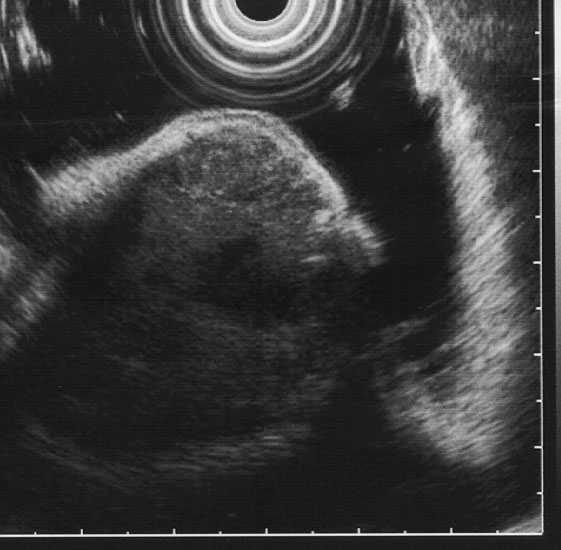
Figure 2. Malignant GIST of the stomach body.
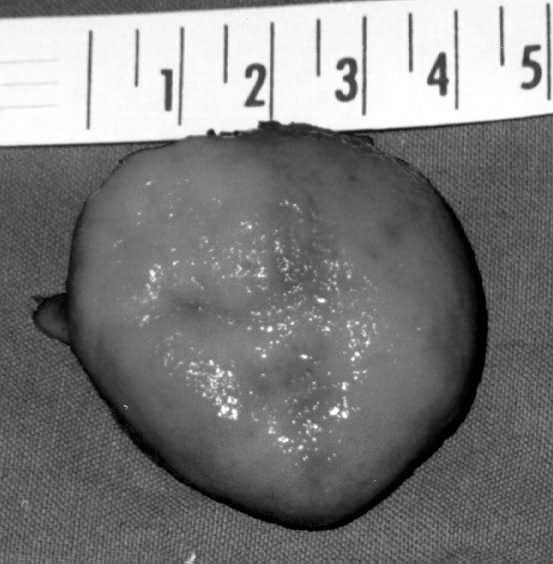
Figure 3. The same tumor as in figure 2 - operative finding.
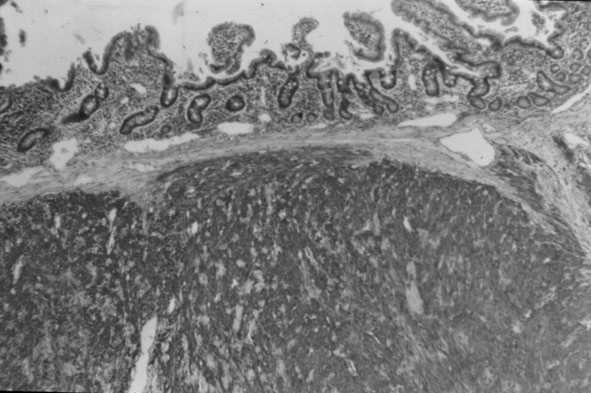
Figure 4. Duodenal benign GIST – immunohistochemistry.

Figure 5. Oesophageal lipoma.
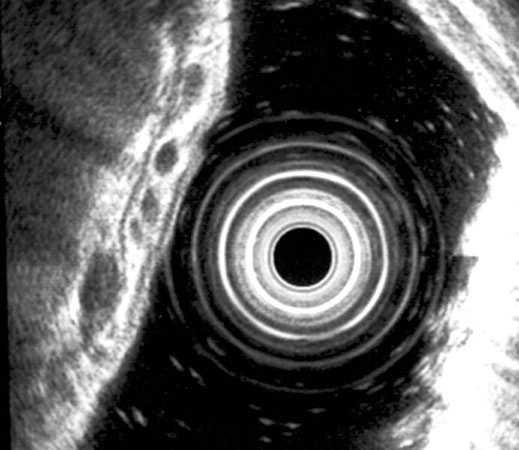
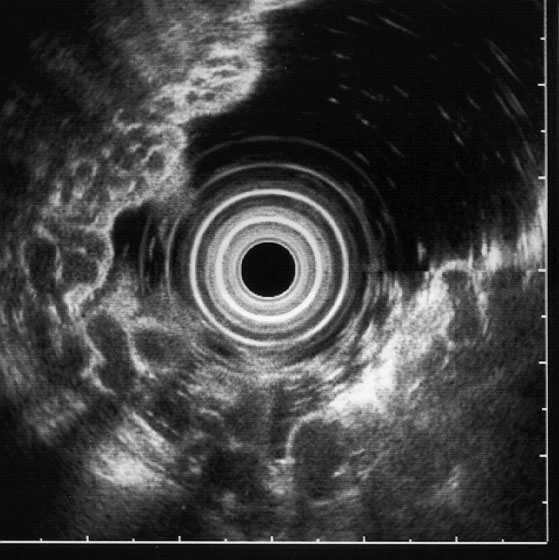
Figure 6a,b. Varices in the stomach fundus.
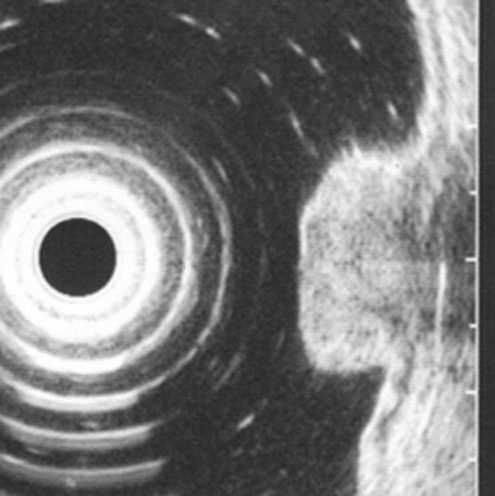
Figure 7. Submucosal metastases of malignant melanom in the stomach.
Table 2. Location of SML in our group patients
Type of lesion | Oesophagus | Stomach Fundus Body Antrum | Duodenum |
| Benign GIST | 9 | 7 | 3 |
| Malignant GIST | 3 2 | ||
| Lipoma | 1 | ||
| Cyst | 2 | ||
| Varices | 4 | ||
| Abberant pancreas | 1 | ||
| Metastases of melanoma | 2 | ||
| Carcinoid | 1 |