Liver and biliary tract
Jetra i bilijarni trakt
Arch Gastroenterohepatology 2000; 19 ( No 3 – 4 )
Management of extrahepatic portal hypertension in children
Lečenje portne hipertenyije u dece
( accepted September 28th, 2000 )
Paediatric portal hypertension
*Vojislav Perišić, Bozina Radević, Dragan Sagić, Dušan Perišić
*University Children , s Hospital, Belgrade, Institute for Cardiovascular Diseases
Dedinje, Belgrade.
Address correspondence to: Profesor Dr Vojislav N. Perišić
University Children , s Hospital
Tirsova 10 St.
YU-11000 Belgrade, Yugoslavia
E-mail: [email protected]
Abbreviations used in this article:
PH, portal hypertension; EHPH, extrahepatic portal hypertension; PVO, portal vein obstruction; PSS,portosystemic shunt
ABSTRACT
Twenty-four consecutive paediatric cases with extrahepatic portal hypertension
( age range, 4 to 15 years ) underwent shunting procedures between July 1995 and April 2000, except one child with complete portal-mesenteric-splenic thrombosis. Type of shunt included: distal splenorenal ( n=21 ), and central splenorenal ( 2 ). At median follow – up of 14 months, ( range 6 months to 5 years ), 21 shunts were patent, 2 were blocked Four patients required readmission due to either anastomotic stenosis ( 3 ) or appositive portal clot grow. Two of them rebled. Variceal bleeding was controlled by endoscopic sclerotherapy in both cases. In 2 patients with shunt stenosis, percutaneous angioplasty was successful in relieving anastomotic obstruction. No patient developed portosystemic encephalopathy. After shunt surgery weight for age and height for age improved in all but two.
It is concluded that portal-systemic shunting is safe and effective method for the treatment of portal hypertension in children. In selected children portal-systemic shunting may become first-choice procedure in experienced centers.
Key words:children, extrahepatic portal hypertension, shunt surgery.
SAZETAK
Dvadesetčetvoro dece sa ekstrahepatičnom portnom hipertenzijom ( uzrastna dob:4 – 15 godina ) je podvrgnuto šant operacijama od Jula 1995 do Aprila 2000 godine, osim deteta sa potpunom portno-mezenterično-lijenalnom trombozom. Vrste šant operacija su bile sledeće: distalni spleno-renalni ( 21 ) i centralni spleno-renalni šant ( 2 ). Tokom praćenja operisane dece ( prosečno 14 meseci; raspon 6 meseci do 5 godina ) u 21 pacijenta šant je bio prolazan i suficijentan, dok je u 2 dece doslo do njegove okluzije. Tokom perioda praćenja 4 dece je ponovo bolnički lečeno zbog stenoze anastomoze ( 3 ) i apozicionog rasta tromba u veni porte ( 1 ). Dvoje je imalo variksno krvarenje. Ono je kontrolisano injekcionom skleroterapijom. U dvoje od 3 dece sa stenozom šanta perkutanom angioplastikom je izvršena uspešna dilatacija anastomoze. Ni u jednog lečenog deteta nije registrovana encefalopatija. Posle uspešno izvedenih operacija uočen je ubrzan rast dece i prirast telesne tezine.
Zaključujemo da su šant operacije uspešan i bezbedan metod lečenja dece sa ekstrahepatičnom portnom hipertenzijom. U jednoj grupi dece to je metod izbora koji moze da se primeni samo u centrima sa iksutvom u lečenju ove vrste pacijenata.
Ključne reči: deca, ekstrahepatična portna hipertenzija, šant operacije.
INTRODUCTION
Extrahepatic portal hypertension ( EHPH ) due to portal vein obstruction ( PVO ) is the most common cause of upper gastrointestinal bleeding in children ( 1 ). Opinions on the primary management of this condition have long been divided between portosystemic shunting and endoscopic injection sclerotherapy/ ligation. Conventional visdom holds that many children with PVO will ultimately outgrow their variceal bleeding if they can be tided over into adulthood by non-operative measures. e.g. variceal sclerotherapy/ligation ( 2 ,3 ). Recurrent variceal bleeding in almost 30% of patient successfully treated by endosopic haemostatic measures and gastric varical bleeding unresponsive to sclerotherapy in 15% of paediatric cases with EHPH questions the contention that by conservative management children ultimately outgrow their variceal bleeding (3,4 ).
In some centers experienced with portal – systemic shunt surgery ( PSS ) multiplicity of major bleeding episodes, severe hypersplenismus, gross splenomegaly, growth failure, necessity for multiple endoscopic variceal hemostatic procedures, presence of fundal varices, patient noncompliance to sclerotherapy, and remoteness from the referral center formed the main indication for shunt surgery ( 3,4,5,6 ). When operated, these children demonstrate high shunt patency rate, low incidence of post – PSS rebleeding, postoperative growth acceleration, and nutritional improvement ( 7 ). This again reinstituted PSS as alternative to repetitive endoscopic haemostatic procedures, which do not interfere with a persistent height deficit and nutritional impairment in children with EHPH.
PATIENTS AND METHODS
A retrospective chart review was performed of all patients with extrahapatic portal hypertension ( EHPH ) who underwent portal-systemic shunt procedures ( PSS ) and splenic surgery at the University Children , s Hospital, Belgrade between July 1995 and April 2000. Data abstracted included: patient , s age at surgery, diagnosis, indication for surgery, type of PSS performed, clinical course and complications, and length of follow-up. Particular attention was focused on growth velocity and weight gain post- PSS, hematological indices, and splenic size.
RESULTS
Clinical characteristic of the 24 children with EHPH demonstrated that age age range was 4 to 15 years ( mean age: 8.5 years ). There were 17 boys and 6 girls. All patients were referred because of gross variceal bleeding manifested by haematemesis and/or melena, and anemia. Three patients had transient mild ascites. Before referral, all patients had at least two variceal bleeding ( range: 1.7 ) requiring blood transfusion. At admission all children had gross splenomegaly and hypersplenism. In two of them enlarged spleen occupied entire left hemiabdomen.
Oesophageal and / or gastric varices were demonstrated all patients by fiberoptic endoscopy. Portal gastropathy was recorded in 6 children. In four cases variceal bleeding was controlled by endoscopic sclerotherapy by one of us ( VNP ). In other children this was made by conventional measured which included blood transfusions, oesophageal variceal tamponade by Blackmore-Sengstaken tube ( 3 ), vasopressin ( 3 ), and somatostatin ( Sandostatin ) therapy ( 3 ).
Angiography performed exclusively by one of us ( DS ) at the Institute for cardiovascular diseases Dedinje was a mainstay of diagnosis demonstrating PVO in all patients. Percutaneous splenoportography was used during the first 3 years of this series, but subsequently PVO was demonstrated by indirect portography after injection of contrast in the superior mesenteric, coeliac, and splenic arteries.
Angiography showed the site of occlusion to be portal vein alone in 21 patients, the portal and splenic veins in 1 patient, and portal, splenic, and adjacent segment of superior mesenteric artery in 1 patient. FIGURE 1. In this children portal vein had been converted into a cavernomatous mass of venous channels. FIGURE 2. One adolescent girl fulfilled the criteria of idiopathic non-cirrhotic portal hypertension ( Bantie , s disease ). In additon, one 5- year old boy with PVO had portal vein agenesis. He had also coeliac disease and atrial septal defect.
Emergency therapy to control variceal haemorrhage, consisting of blood transfusions, intravenous vasopressin or somatostatin, and / or endoscopic sclerotherapy ( 4 children ), and oesophageal balloon tamponade ( 3 children ) was uniformly successful in this goup of patients. Repeated variceal regrowth necessitating frequent admissions and sclerotherapy sessions under anesthesia and hospital stay averaging 3 days in 5 children was important indication for shunt surgery. Two children were operated because of gastric variceal and portal gastropathy bleed.
Partial splenectomy ( lower lobe resection ) was definitive treatment in child with complete portal-mesenteric-splenic thrombosis, massive splenomegaly, and severe hypersplenismus. Interestingly this child did not have oesophagel varices. On the contrary portal gastropathy and gastric varices of grade I was present. Figure 2. After partial splenic resection performed by one of us ( BR ) all haematological indices returned to normal; growth velocity increase and weight gain were registered.
Distal side – to – side splenorenal shunt without splenectomy was performed in 21 patients by one of us ( BR ). Central end – to – side splenorenal shunt with splenectomy was done in 2 patients by the same surgeon ( BR ). The choice of this type shunt was based on the enormous splenic enlargement, severe hypersplenismus, availability and size of veins. A large wedge-shaped specimen of the liver was obtained from all patients, and biopsy results confirmed the absence of liver disease in each operated child. After operation, significant decrease in splenic size and quick amelioration of haematological abnormalities due to hypersplenism
Shunt patency was determined during follow-up by angiography and/ or Doppler ultrasonography. In four patients, all with distal side-to-side splenorenal shunts, shunt occlusion occurred. Occlusion of the shunt was discovered within one year after the operation in all children. In the first 3 cases percutaneous shunt angioplasty was performed by one of us ( DS ). Obstruction was relieved in 2 children. In the fourth case, appositive portal vein clot grow, further obstructed portal and splenic vein thus unabling any angiographic interventional and surgical procedure.
In two children, recurrent variceal bleeding occurred postoperatively. These were patients who had occluded spleno-renal shunts. After sclerotherapy, diagnostic angiography was performed and showed occlusion of the shunt. Immediate shunt angiopasty was made during each procedure.
After PSS, general health, quality of life, and child catch up growth was judged excellent or good in 23 cases. Children are engaged all children play without any limitation and going to school regularly.
DISCUSSION
The result described in this report indicates that portal-systemic shunt ( PSS ) is a highly effective and long lasting treatment modality for EHPH caused by PVO ( 4,5,6,7 ). The morbidity of PSS was very low. There were no operative deaths, and survival was 100%. All but two shunts remained patent throughout the period of observation, a patency rate was 83%. Thus, PSS was successful in 83% of patients. Growth, development, and quality of life were generally very good throughout the period of observation. Liver function and histology remained normal.
Treatment of children with bleeding varices in EHPH due to PVO has changes with the development of endoscopic therapies in eighties ( 3,8,9 ) . Endoscopic variceal sclerotherapy and variceal band ligation became the initial therapy for bleeding oesophageal varices in childhood ( 3,7,9,10 ) This led to abandonment of shunt surgery in the majority of paediatric departments. Sclerotherapy has become the sandard care of children with EHPH and bleeding oesophageal/gastric varices ( 3,7,9,10 ). In a light of the good result with endoscopic methods combined with the natural history of PVO and naturally developing collaterals, one may ask whether shunt surgery has any place in the treatment of EHPH?
The good results of PSS in this series are in concordance with reports from experienced centers with surgery of EHPH. Bismuth reported shunt thrombosis in only 5%, Alvarez in 8%, Mitra in 13% ( 3,6,11 ). This may indicate that in properly selected cases the majority of operated children with EHPH may experience permanent shunt patency without requiring repetitive endoscopic treatments and hospitalisations ( 7 ). Therefore, some centers accepted the alternative of PSS under elective circumstances early in the course of treatment of children with bleeding varices due to EHPH. The rationale was that PSS, but not endoscopic therapy permanently decompress the portal system.
There is common belief that children with EHPH due to PVO and oesophageal/gastric varices should no be referred to PSS because one may expect that episodes of variceal bleeding decrease and ultimately subside in many children as they grow older ( 12 ). But, there is little doubt that some, if not most paediatric patients with EHPH will continue to have episodes of variceal haemorrhage until they receive effective therapy ( 4 ). Long-term endoscopic therapy has had failure rate exceeding 20% thus necessitating repetitive endoscopic haemostatic sessions and hospitalisations ( 3 ). Thus our group adopted a policy of PSS in the absence of failed endoscopic control of variceal haemorrhage what was already reported by Tzakis group from Miami ( 7 ). Endoscopic control of actual variceal haemorrhage combined with PSS led to high rate of variceal disengagement which parallels achieved portal decompression.
We preferentially performed distal spleno – renal shunt because it has a low incidence of portosystemic encephalopathy, and splenectomy is avoided. Our experience confirmed the experience by Bismuth, Alvarez, Mitra, and Tzakis that the size of the anastomosis in the youngest patients did not play a role in the incidence of thrombosis or stenosis when proper surgical technique was applied by experienced surgeon( 11,3,6,7 ). Fear of the development of thrombosis in small vascular anastomosis was no confirmed by aforementioned authors.
Preoperative angiography was important in delineating lienal, mesentyeric, and portal veins morphology and in anticipating potential surgical problems ( 13 . Splenic vein thrombosis due to extension of the cavernous change in the portal vein was important to detect prior PSS. Routine use of postoperative angiography was not necessary. We found that ultrasound examination by Doppler technique was enough sensitive to assess shunt patency and measure quantitatively anastomotic blood flow.
We further demonstrated that operated children with EHPH improve their growth and weight gain after PSS. This further confirmed already published observations by Alvarez, Sarin, and Tzakis group ( 3,4,6 ). The precise cause of this phenomena is unknown. This observations was not recorded in children with EHPH with bleeding varices obliterated by sclerotherapy, because endoscopic therapy does no decompress the portal system.
We conclude that elective PSS in selected children with EHPH due to PVO is first line treatment. This may apply in patients with intrahepatic portal hypertension with preserved liver function tests who are not immediate candidates for liver transplantation. In order to control actual variceal bleeding and prepare children with EHPH for shunt surgery, paediatric endoscopic services have to be available in parallel.
REFERENCES:
1.Perisic VN. Portna hipertenzija u dece. U: Radevic B, Perisic VN, Sagic D ( eds ). Portna hipertenzija. Beograd: Zavet, 1999; 81 – 3.
2.Fonkalsrud EW, Myers NA, Robinson MJ. Management of extrahepatic portal hypertension. Ann Surg 1974; 180: 487 – 93.
3.Stringer MD, Howards ER. Long term outcome after injection sclerotherapy for oesophageal varices in children with extrahepatic portal hypertension. Gut 1994; 35: 257 – 9.
4.Orloff MJ, Orloff MS, Rambotti M. Treatment of bleeding esophagogastric varices due to extrahepatic portal hypertension: Results of portal – systemic shunts during 35 years. J Pediatr Surg 1994; 29: 142 – 54.
5.Alvarez F, Bernard O, Brunelle F, Hadouchel P, Odievre M. Portal obstruction in children. Part II: Results of surgical porto – systemic shundting. J Pediatr 1983; 183: 703 – 7.
6.Mitra SK, Rao KL, Narasimhan KL ey al. Side – to – side lienorenal shunt without splenectomy in noncirrhotic portal hypertension in children. J Pediatr Surg 1993; 28: 398 – 432.
7.Kato T, Romero R, Mittal NK et al. Portosystemic shunting in children during the era of endoscopic therapy: improved postoperative growth parameters. J Pedfiatr Gastroenterol Nutr 2000; 30: 419 – 25.
8.Hasal E, Berquist WE, Ament ME, Vargas J. Sclerotherapy for extrahepatic portal hypertension. J Pediatr 1989; 115: 69 – 74.
9.Perisic VN. Endoskopska injekciona skleroterapija variksa jednjaka u dece. Gastroenterohepatol Arhiv 1985; 4: 63 – 6.
10.Price MR, Sartorelli KH, Karver FM, Narkewicz MR, Sokol KJ, Lilly JR. Management of esophageal varices in children by endoscopic variceal ligation. J Pediatr Surg 1996; 31: 1056 – 9.
11.Bismuth H, Franco D, Alagille D. Portal diversion for portal hypertension in children, the first ninety patients. Ann Surg 1980; 192: 18 – 24.
12.Webb LJ, Sherlock S. The aetiology, presentation, and natural history of extra – hepatic portal venous obstruction. Q J Med 1979; 48: 627 – 39.
13.Sagic D. Dijagnoza i interventne procedure u portnoj hipertenziji. U: Radevic B, Sagic D ( eds ). Portna hipertenzija. Beograd: Zavet, 1997; 49 – 70.
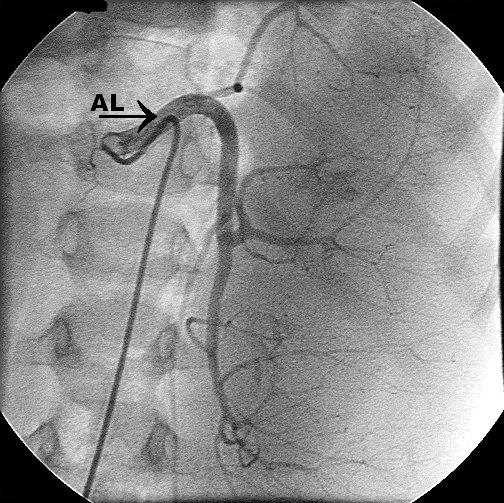
Figure 1. Site of portal vein thrombosis/obstruction in 22 children with portal vein obstruction. PV: portal vein. SMV superior mesenteric vein. SV: splenic vein.
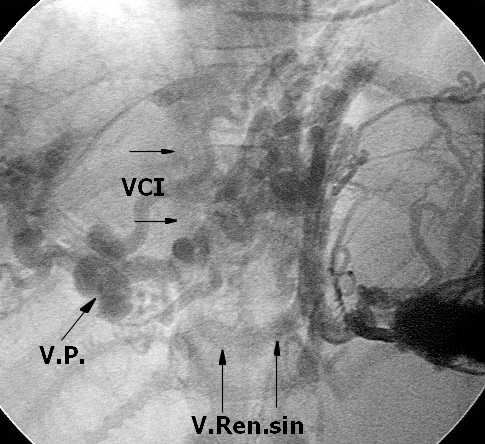
Figure 2a. Splenoportography. Note cavernomatous nsformation of portal vein ( VP ) and splenic vein. Quick opacification of left renal vein ( V.Ren.sin ) and inferior v.cava ( VCI ) indicates the presence of spotaneous spleno-renal collateral(s).
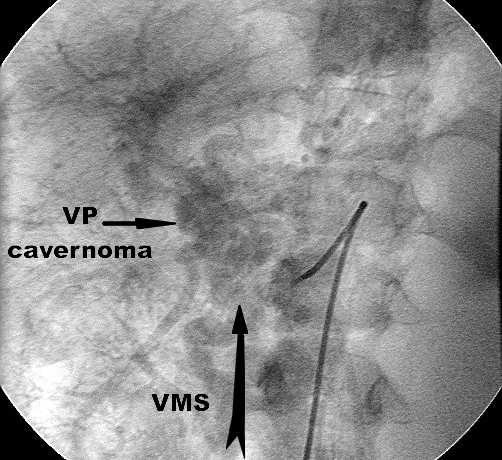
Figure 2b. Indirect superior mesenteric venography. Near the junction with portal vein, superior mesenteric vein ( VMS ) is obstructed ( arrow ) and converted into cavernomatous mesh.
Concluson: Portal hypertension due to total splanchnic ( portal-mesenteric-splenic ) thrombosis.