 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
|
Back to Introduction page |
|
| Structure of Carbon Fiber |
|
|
|
|
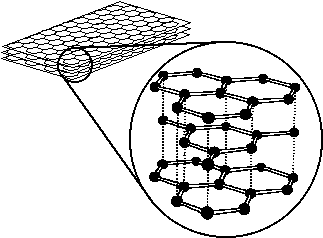
| The structure of carbon fiber is very similar to that of ordinary graphite (see figures 1 and 2) in that it is made up of sheets of carbon (graphite) formed by hexagonal rings of carbon atoms (see figures below). |
|
|
|
 |
|
|
| Figure 1: the structure of graphite. |
|
|
| Each 'ring' is made up of five single bonds and one double bond, so that while each carbon atom is only bonded to three others, it is still sharing enough electrons to have a stable octet. |
|
|
| As you can see the sheets of carbon in the graphite are aligned parallel to each other, because of this the bonds between the sheets can be broken fairly easily, letting the sheets 'slide' across each other. When you are writing with a pencil the 'lead' is actually made up of mostly graphite and as you drag the tip of the pencil across paper 'sheets' of graphite are actually sloughing off onto the paper forming a visible line. Because of this, graphite is also an excellent lubricant. |
|
|
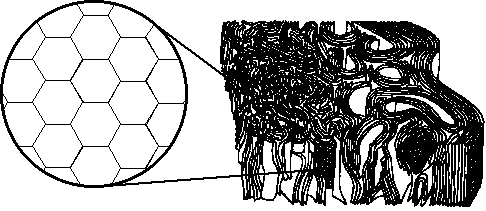
| When graphite is in the form of carbon fiber, these sheets are 'rolled' together to form strands or threads. (See figures 2 and 3). |
|
|
 |
|
|
| Figure 2: The structure of carbon fiber. |
|
|
| Here you can see how the graphite sheets are sort of tangled together to form a single strand or filament. These strands are most often used in bunches or threads, and are classified by the number of strands in each thread (usually 2-20 thousand). |
|
|
| Below is a picture of carbon fiber 'thread'. |
|
|
 |
|
|
| Figure 3: A spool of carbon fiber thread. |
|
|
| The reason that carbon fiber thread does not just pull apart (let the graphite sheets slide past each other), is that they (the carbon sheets) are also interlocked with each other as shown in figure 4. |
|
|
 |
|
|
| Figure 4: The inter-locking graphite sheets of carbon. |
|
|
| Images from |
|
|
| http://www.carb.com/carbon_fibers.html |
|
| Information from the same. |
|







