

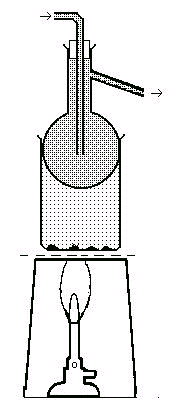
Ok, lets see an easy experiment supposing you want to prepare the Iodine first:
You will need:
Bunsen burner and asbestos square (or a hot plate) Tripod and tin lid Beaker, 250 ml Separating funnel 100ml Evaporating dish Hydrogen peroxide 20 volume Tetrachloromethane or dichloromethane Distilled water 1M solution of Sulphuric acid Supply of ribbon seaweed(Laminaria) - that you can obtain a a biological supply store or from the sea shore Procedure (You must work at a fume cupboard) 1-Collect and dry about a dozen 50cm lenghts of the seaweed and heat them strongly on a tin recipient until they are reduced to ash; probably it will reduce a quite small quantity, about a spoonful. 2- Add to the ash 20 ml of distilled water in a beaker, and heat the suspension until it boils. 3 - Filter the suspension 4 - Acidify the filtered with Sulphuric acid solution 5 - Add then the hydrogen peroxide solution You will observe the formation of a brown color due the iodine liberation from the iodine ions present 6- Transfer the mixture to a separating funnel 7 -Extract the Iodine with tetrachloro methane or other solvent. The result is an organic solution of iodine. 8 - In order to obtain crystals , the solvent may be allowed to evaporate at room temperature, by placing it in an evaporating dish in a fume cupboard. It will result crystals of iodine or rather gray- black brilliant flakes.






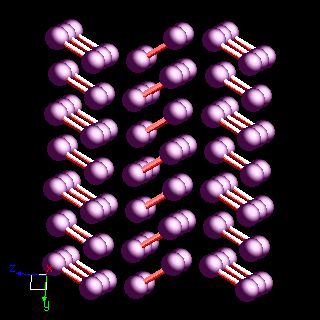
CRYSTALLINE STRUCTURE OF IODINE
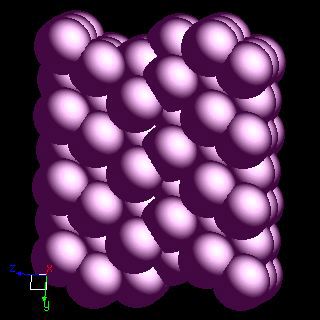
ORBITAL STRUCTURE OF IODINE
