Chapter 2 : Hardening of the cement paste
Hydration of cement:
Hydration is the term used
to describe the reaction between the cement powder and water. Hydration takes
place on the surface of the cement grain. As hydration progresses, heat is
generated (hydration is a
exothermic reaction) and the grains get coated by solid products called
hydrates. Smaller grains of cement hydrate first and are the first to be
completely hydrated. While larger grains becomes smaller with time and some may
never hydrates.
The hydration reaction will continue as long as heat and moisture are present. The cement components will react in the same time but at different rates. The hydration reaction requires a little amount of water. The additional water in the concrete is needed for the workability.
Components of the cement:
|
C3S |
3CaO.SiO2 |
Tricalcium silicate |
50% |
|
||
|
C2S |
2CaO.SiO2 |
Dicalcium silicate |
25% |
|
||
|
C3A |
3CaO.Al2O3 |
Tricalcium aluminate |
10% |
|||
|
C4AF |
4CaO.Al2O3.Fe2O3 |
Tetracalcium aluminoferrite |
10% |
|||
|
CSH2 |
CaO.SiO2.H2O |
Gypsum (calcium Sulphate)
|
5% |
|||
Where: CaO=C SiO2=S Al2O3=A Fe2O3=F H2O=water
Chemical reactions:
Hydration of C3S and C2S:
C3S + H2O = C-S-H
(Calcium Silicate hydrate) + CH (calcium Hydroxide) + Heat
C-H-S is amorphous and fibrous, makes up 50-60% of the solids in the hardened paste and it forms a continuous binding matrix with a large surface area and it is the component responsible for the development of strength in the cement paste.
CH is a crystalline, thick
hexagonal plate. Imbedded in the C-H-S matrix filling the pores, CH is not
important in the strength of the hardened paste.
The hydration of C2S is the
same as the hydration of C3S which produce the same hydrates with similar
properties but the reaction of C2S is slower and generates less heat because it
is less active therefore it contributes to the later strength rather than the
early strength like C3S does.
Hydration of C3A and
C4AF:
C3A reacts with water in a very violent and rapid way leading to flash setting,
C3A + H2O = Calcium aluminate hydrate (C-A-H) + Heat
C-A-H forms bridges of solid materials between the cement particles. that’s the reason why gypsum is added to the cement to produce Ettringite (calcium sulphoaluminate), C3A hydrates with sulfate ions supplied by dissolved gypsum:
C3A + Gypsum +
water = Ettringite (Trisulfate hydrate)+ Heat
If the gypsum is too little, it will react with C3A too vigorously and flash setting will occur. If the gypsum is too much an expansive reaction will take place.
Ettringite is a long, slender, prismatic, crystalline, needle like material that makes up for 10-20% of the solids in the paste. And its stable as long as long as gypsum is present. It plays a minor role in strength but a major one in durability.
C3A
will continue to react with gypsum till all the gypsum is consumed then C3A
will start to react with Ettringite to form Monosulfate, which will set the
paste.
C3A + Ettringite
+ water = Monosulfoaluminate hydrate + Heat
Monosulfoaluminate a
stable hydration product which is a thin, irregular plate, clustered like
flower petals and fairly crystalline. It fills the pores but can be reformed
into ettringite if sulfates ions are able to enter the concrete (Sulphate
attack). This new formation causes volume to increase and leads to tensile
cracking. This tendency is the basis for sulfate attack of Portland cements.
Rate of hydration:
The hydration rate has the following order: C3A>C3S>C4AF>C2S
Ettringite forms first, followed by CH and C-S-H and a little change in the
total volume of the paste is a result of the hydration.
The rate of hydration can
be increased in the following ways:
-Temperature: for each 10C increase in temperature, the rate of hydration doubles.
-Fineness of cement: the more the fineness of the cement the more the surface area that the -hydration can take place at.
-Changing the proportion of
the cement components, like increasing C3S and C3A.
The final product of hydration is a hardened paste
consists of solids (C-H-S, CH, Ettringite, Monosulphate, unhydrated cement) ,
water, air voids and capillary voids.
Heat of hydration:
During the hydration process, heat is liberated.
Around 30% of the total heat is generated within the first day. And around 50%
within the first three days and since the thermal conductivity of concrete is
low. It acts as an insulator containing the heat with in the concrete itself
forcing the temperature to rise up to 50C which may cause cracking.
Adjusting the fineness and composition of cement can lower the heat of hydration. Decreasing C3A and C3S as in type IV cement will lower the heat evolution during hydration.
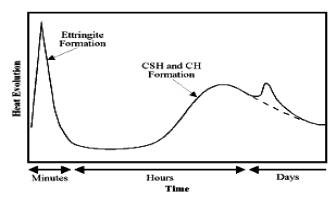
Hydration stages:
The actual process of cement
hydration, for the purposes of modeling the development of microstructure, can
be broken down into three parts:
1)
Material dissolves from the original
cement particle surfaces.
2)
Diffuses within the available pore
space.
3)
Ultimately reacts with water and other
dissolved or solid species to form hydration products through aggregation.
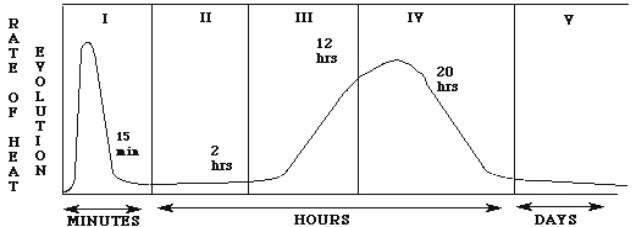
Stage I : Initial stage
(0-15 minutes)
Immediately after mixing
the cement powder and water together to form a gel-like viscous liquid.
Cement Gains of different
sizes hang in the water solution.
Materials start to
dissolve from the surface of the grains and defuse into ions.
The Sulphate ions (from
the gypsum) react with the aluminate ions (from C3A) to form Ettringite as very
fine-grained crystals in the solution, which are very small to form bridges
between the cement grains so the cement paste remains plastic
Ettringite starts to
increase in the solution forming a gel coating all grains
Some of the ettringite in
the solution crystallize as needle like crystals out side this layer.
The rate of dissolving and
diffusion of ions on the grains surface decrease as the ettringite gel gets
thicker until no water can penetrate the gel to reach the grain surface to
dissolve more materials and diffuse more ions.
Stage II : Dormancy
stage (15 min-4 hours)
Because the solution
around the gel is supersaturated with the ions the diffusion of the ions stop
and the fact that the ettringite gel is very thick now stops the water from
penetrating the gel to reach the surface of the grain.
Because some water already
exists between the water and the grain some materials dissolve and diffuse
behind the gel barrier and with time the ions concentration between both sides
of the ettringite gel causes osmosis pressure that finally rupture the gel.
Stage III :
Acceleration stage (4-8 hours)
After the ettringite
membrane is ruptured, water reach the surface of the grain dissolving more
materials and diffusing them into ions and because the solution around the
grain is saturated , hydration products of the ions starts to precipitation on
the grain. Decreeing the concentration of the ions and triggering more ion
diffusion of the different four grains of cement. The dissolving of the grain
surface and the ions release starts again increasing the heat generated.
The same happens with the
C3A grain. As more ettringite is formed in the needle crystals. These needles
interlock with other on the adjacent grains causing the cement to set (initial
set)
Stage IV : Deceleration
stage (8-24 hours)
Because of the hydration
products building up by the minute, the water faces more difficulties in
reaching the surface of the grain in order to dissolve more materials resulting
in a decrease in the generated heat.
Stage V : Steady stage.
With time, water is facing
more thickness to reach the grain through the hydrates pores; the reaction
slows down but continues as long as there is water available for the ions to
dissolve.

Setting of cement paste:
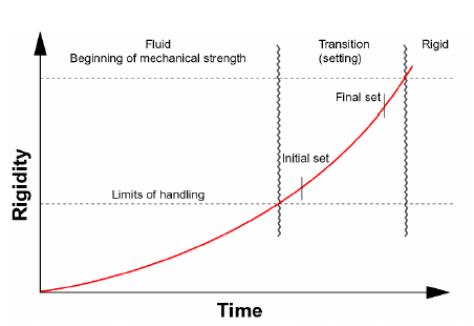
Setting is defined as the onset of rigidity in fresh concrete. Hardening is the development of useable and measurable strength; setting precedes hardening. Both are gradual changes controlled by hydration. Fresh concrete will lose measurable slump before initial set and measurable strength will be achieved after final set.
Setting is controlled by the hydration of C3S. The period of good workability is during the dormant period, (stage 2). Initial set corresponds to the beginning of stage 3, a period of rapid hydration. Final set is the midpoint of this acceleration phase. A rapid increase in temperature is associated with stage 3 hydration, with a maximum rate at final set.
If large amounts of ettringite
rapidly form from C3A hydration, the setting times will be reduced. Cements
with high percentages of C3A, such as expansive or set-regulated cements, are
entirely controlled by ettringite formation.
Up normal setting behavior:
False Set : Early stiffening of concrete, fluidity may be restored by remixing.
Basically, it is a result of hydration of dehydrated gypsum, which forms rigid
crystals. Because there are few of these crystals and they are weak, the matrix
can be destroyed by remixing. Accelerated hydration of C3A will cause rapid
development of ettringite and false set.
Flash Set : Stiffening of concrete due to the rapid development of large quantities of C3A hydration products which cannot be returned to a fluid state with mixing. This is generally no longer a problem since the introduction of gypsum to control C3A hydration. However, some admixtures will increase C3A hydration and flash set may be a problem.
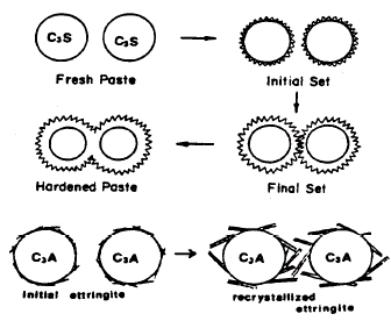
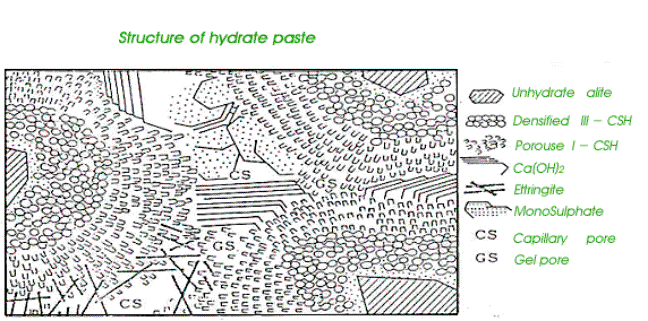
Paste microstructure:
When C3S hydrates it needs to grow in free spaces or the growth will stop. It starts to produce C-H-S coatings on the surface in very little quantities in stage (II) of hydration then increase rapidly in stage (III). The growing spines (cones) of the C-H-S radiate outward from the C3S particle with the bulk hydrates on the bottom. As the coating get thicker the adjacent spines from two different cement grains are forced to interlock to form solid bonds. Or the spines could bond to aggregates.
The CH only grows in free capillary spaces if blocked it will change direction and could grow around the cement grain with no effect on strength
Ettringite will grow capillary pores too and later reformed to monosulfoaluminate but some will never hydrates, as CH will cover it.
Porosity:
Water is required for
cement to react and bind the concrete components into a solid mass. The
theoretical water/cement ratio required for full hydration of the cement
is (0.3). However this would produce unworkable
concrete. So additional water is needed. This excess water determines the
properties of concrete : strength, workability and durability.
When the cement and water
are mixed, a cement-water gel is formed and with time, parts of this gel start
to hydrate producing solid products. At full hydration most of the cement is a
solid product and the mix water takes one of three forms: combined water, gel
water and capillary water.
Some water is combined
chemically or physically with the products of hydration, and is held very
firmly this water is called “combined water” (23% of the mass of the dry
cement)
The hardened paste consists of the solid products of
hydration and water held physically or absorbed on the surface area of the
hydrates. This water is called “gel water” as it is held in the cement gel. And
it is located between the solid products of hydration (C-H-S) in pores called
“gel pores” (2 nm in diameter and about 28% of the cement gel) because these
pores are very small they are impermeable.
The excess water not used in hydration is stored in capillary pores which is larger than the gel and combined water pores (1micrometer and about 18.5% of the volume of dry cement) after the hydration of cement these capillary pores can be empty or filled with water. If the mix contained more water than necessary for full hydration, there will be more than 18%, which are all full with water
While the paste is still hydrating the capillary pores are connected which each other by channels but if the degree of hydration is sufficiently high, this inter connecting system becomes segmented and blocked by newly hydrated paste lowering the permeability but the capillary pores are still partially connected.
Whenever concrete is mixed using normal mixers, air will be entrapped as bubbles although compaction will release most of it. But air bubbles will still be entrapped.
Once the paste has set it gets hardened and it’s gross volume remains approximately constant. And the concrete is formed of aggregates, hydrated products and pores. These pores consist of the air bubbles entrapped and the capillary pores that used to contain the excess water (the water added for workability) the excess water maybe evaporated or some is still in these pores.
The porosity of the concrete is the volume of the air bubbles and the capillary pores. Where the permeability of the concrete rises from the channels that connect the capillary pores. So the paste could be high in porosity (high excess water like in higher w/c) but low in permeability (high rate of hydration and well curing) and the opposite is also possible. While some concrete could be high in both porosity and permeability.
The curing of the fresh concrete plays a major role in permeability, as stopping the curing for a while will cause the paste to shrink and cracks will form increasing the permeability.
Development of strength:
The strength of hardened concrete depends on the total volume of voids (porosity plus entrapped air) independent on the fact that the capillary voids are full of water or empty.
A fine bond crack forms at
the interface of the coarse aggregate and the hardened paste due to
differential volumetric changes between the paste and the coarse aggregates.
When the concrete is loaded at stresses above 30% of the ultimate stress, the
microcracks starts to increase in length, width and number. At 70-90% of the
ultimate strength, cracks open through the mortar (paste plus fine aggregate)
and connects the bond cracks so a continues crack pattern is formed. If the
load is kept, failure will occur with time. If the load is increased, rapid
failure will take place at maximum stress.
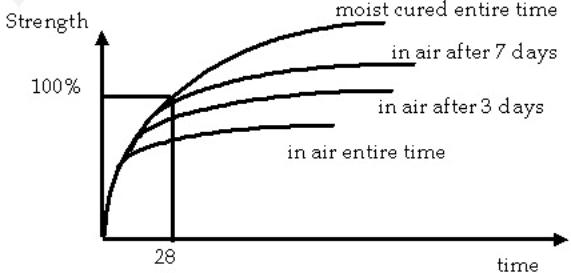
Curing:
Curing is the process used for enhancing the hydration of the cement paste by controlling the temperature and moisture. The object of curing is to keep the concrete saturated as much as possible until the water filled voids are occupied with the products of hydration.
The hydration of cement can
take place only in free voids (only the water voids are free) If the internal
relative humidity drops below 80%, hydration and strength gain will stop. The
rate of strength gain is directly related to the amount of moist curing. The
loss of this water by evaporation or absorption by the aggregates, formwork, or
subgrade must be prevented. The curing should continue until all the capillary
voids in the paste has been segmented and blocked
Evaporation above 0.5 Kg/m² per hour should be prevented while the evaporation of the water depends of the temperature, relative humidity and wind velocity.
If the curing is interrupted which is more of a problem during early hydration than later in the concrete's lifetime. The wetting and drying can cause the concrete to be susceptible to tensile cracking developed during drying.
if the relative humidity falls below 80%, hydration will stop. A fully saturated concrete will be able to provide water to localized areas in the paste that are starved for moisture. Concrete that is sealed against moisture loss will hydrate and gain strength more slowly than a continuously moist cured concrete.
Increased temperature results in
improved early strength and lower ultimate strength. The early strength gain is
explained by the increase of the hydration process. The lower ultimate strength
is more difficult to explain, but seems to be related to non-uniform development
of the microstructure. ACI
suggests that 7 days of moist curing, or the time necessary to attain 70% of
the specified compressive strength, whichever is less, is adequate for
structural concrete. Non-reinforced concrete requires longer times. At
temperatures below 0 C, freezing can be a problem. The concrete should not be
allowed to freeze until it has developed some strength (500 lb/in2).