Isomerism is the phenomenon which certain compounds processing the same molecular formula exist in different forms due to their different arrangement.
Isomerism can be separated in different categories. The particular types of isomers which are different from each other only in the way the atoms are oriented in space are known as stereoisomers.
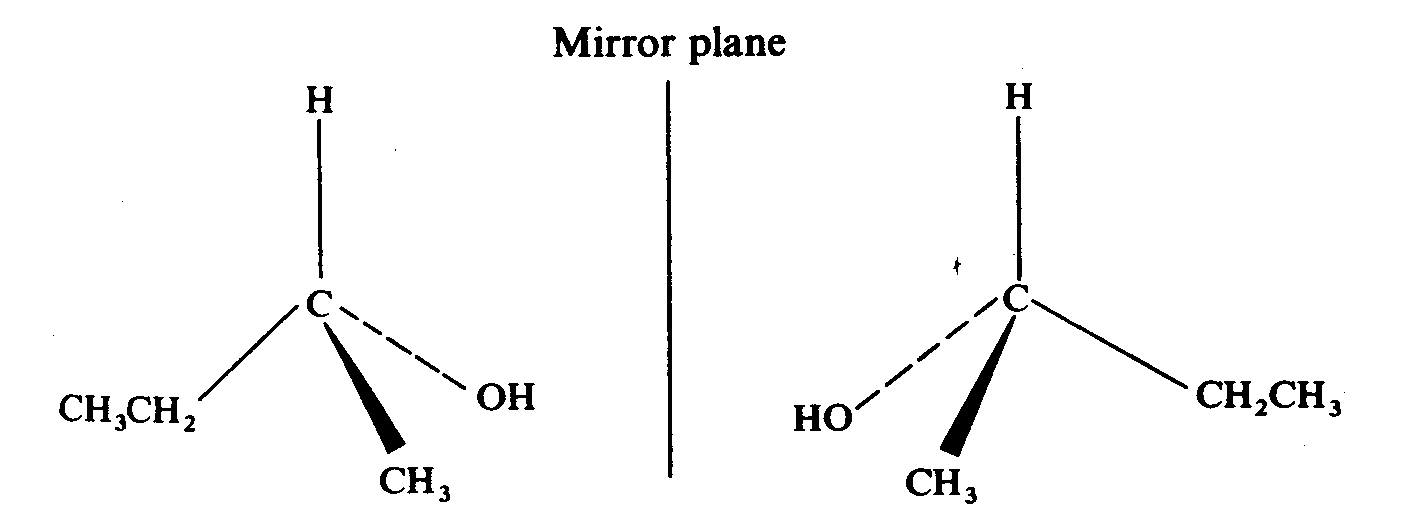
Enantiomer is one type of stereoisomer. Take butan-2-ol as an example. We find that there are two possible ways to draw it.

They are mirror images of one another and they are not superimposable. This is known as enantiomerism and the isomers are called enantiomers.
Other examples include:
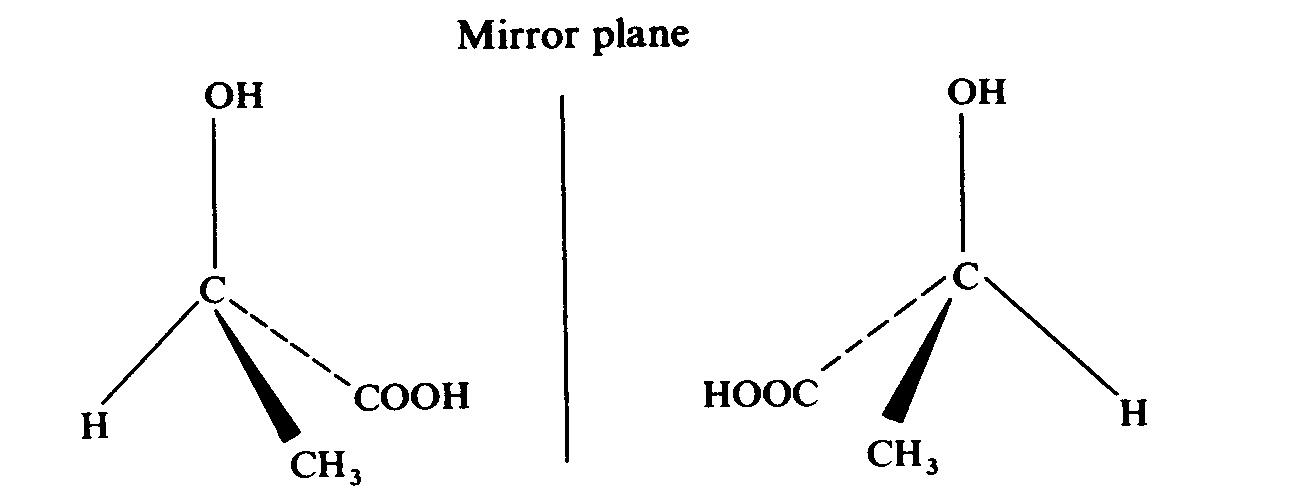
2-hydroxypropanoic acid (lactic acid)
2-amainopropanoic acid (alanine)
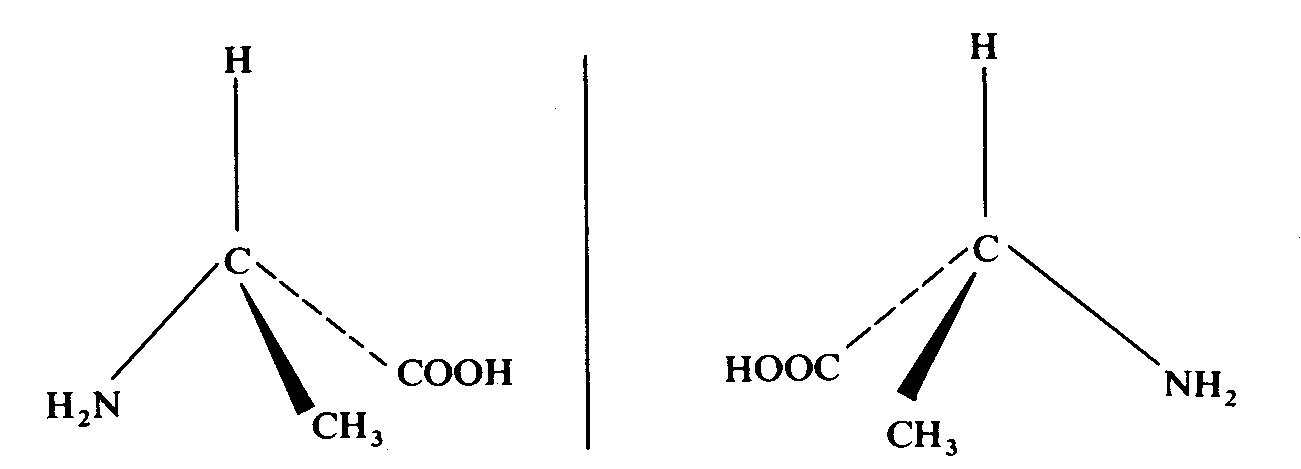
Tale a closer look on the compound. We find that they all have one thing in common --- a carbon atom is attaching to four different groups. The carbon atom is known as a chiral centre. For example, for butan-2-ol, the four groups attaching to the chiral centre are methyl group, ethyl group, hydrogen atom and hydroxy group.
Enantiomers have identical melting point, boiling point, density and refractive index. However, their optical activity are different, i.e. their degree of rotation of polarized light are different. The direction of rotation of polarized light in a pair of enantiomers are always opposite and have the same magnitude.
For example, for 2-methylbutanol, the specific rotation
Moreover, enantiomers have same chemical properties towards optically inactive compounds but different chemical properties towards optically active compounds.
There are different ways to differentiate two enantiomers. One way is by using Cahn-Ingold-Prelog sequence:
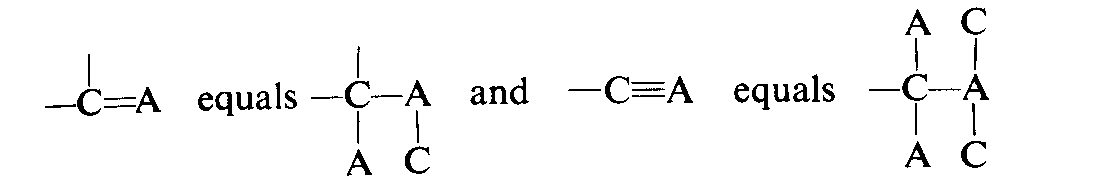
For example,
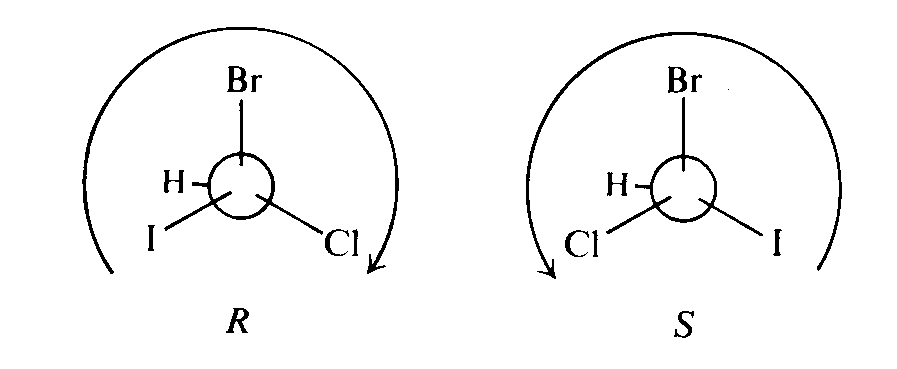
If equal amount of enantiomer is mixed together, the specific rotation of the polarized light on the sample will become zero. As a result, the optical activity of both enantiomers is said to be externally compensated. The mixture is called a racemic mixture.
There are other type of steoreoisomer known as diastereoisomers. Diastereoisomers are stereoisomers which are not enantiomers. For example,
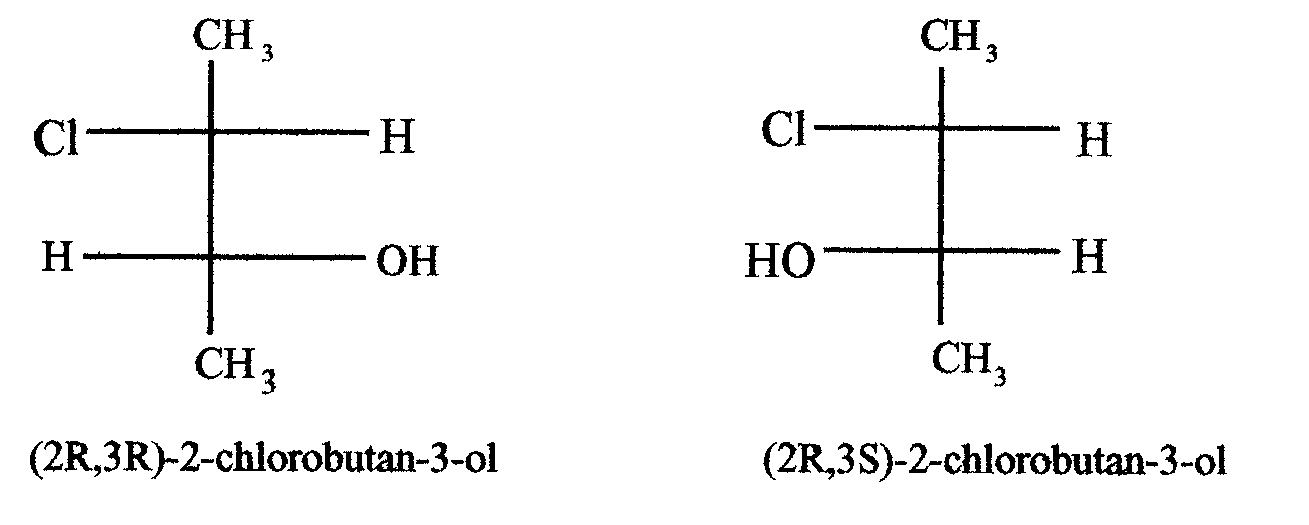
Take a look on 2,3-dichlorobutane. It is obvious that (R,R)-isomer and (S,S)-isomer are enantiomers. However, its (R,S)-isomer and (S,R)-isomer are identical. Indeed there is a plane of symmetry between the second and third carbon. Thus, the compound is optically inactive. This is called a meso compound and we said that its optical activity is internally compensated.