|
[Logo CDER] CHEMISTRY REVIEW
TEMPLATE [Logo CDER] |
Chemistry Assessment Section
a. Proprietary Name: Wellbutrin XLTM
b. Non-Proprietary Name / USAN: bupropion hydrochloride
c. Code Name/# (ONDC only): N/A
d. Chem. Type/Submission Priority (ONDC only):
· Chem. Type: 3
· Submission Priority: S
9.
LEGAL
BASIS FOR SUBMISSION: 505(b)(1)
10. PHAMACOL. CATEGORY:
Treatment of major depressive disorder
11. DOSAGE FORM: Extended Release Tablets
12. STRENGTH/POTENCY: 150 mg and
300 mg
13. ROUTE OF ADMINISTRATION:
oral
14. Rx/OTC DISPENSED: _ X _ Rx _OTC
15. SPOTS (SPECIAL PRODUCTS
ON-LINE TRACKING SYSTEM):
____ SPOTS product _ Form Completed
_X__ Not a SPOTS product
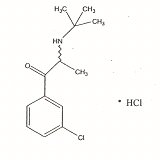
16. CHEMICAL NAME, STRUCTURAL
FORMULA, MOLECULAR FORMULA, MOLECULAR WEIGHT:
Chemical
Name: (±)-2-(tert-butylamino)-3’-chloropropiophenone hydrochloride
Molecular
Formula: C13H18C1NO·HCl
Molecular
Weight: 276.20
17. RELATED/SUPPORTING
DOCUMENTS:
Page 4 of 10
Back a Page
Next Page
Back to Wellbutrin XL NDA Index Page
Back to Main Index Page