NDA
20-358: WELLBUTRIN® SR tablets
structural
formula is shown below:
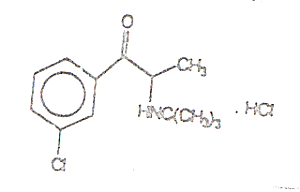
INDICATION
AND USAGE: It is proposed by the firm that
WELLBUTRIN® SR tablets will have the same indication that appears in the
current approved labeling for the immediate release tablet. Bupropion is used orally for the
treatment of depression.
HOW
SUPPLIED:
WELLBUTRIN® SR will be supplied for oral administration as 50 mg (white), 100
mg (blue), and 150 mg (purple) film-coated tablets.
MANUFACTURER
AND MANUFACTURING SITE: The pivotal biobatches
for the three strengths of the SR tablets were made by Burroughs Wellcome
Company in Greenville, N.C.
Further, manufacturing of these three strengths will also occur at the
Greenville facility.
RECOMMENDED
DOSAGE AND ADMINISTRATION (FIRM’S): The
usual initial adult dose is 150 mg/day, given once daily. Patients who are not responding to a
dose of 150 mg/day may benefit from dose increases up to a maximum of 300
mg/day. dose increases should
occur at intervals of at least one week.
Single doses of WELLBUTRIN® SR should not exceed 150 mg. Doses of WELLBUTRIN® SR greater than
150 mg/day should be administered twice a day, preferably with at least 8 hours
between successive doses.
In support
of this submission, the sponsor has submitted 6 studies – 3 pilot or background
and 3 pivotal bioequivalency studies.
An
Overview of Pharmacokinetics of Bupropion (Original Submission, IR tablets)
After
oral administration of WELLBUTRIN® IR tablets, peak plasma bupropion
concentrations are usually achieved within 2 hours. Mean elimination half-life of bupropion following single-doses
oral administration ranged from 9-14 hours. Oral clearance is approximately 2.0 L/hr/kg. Plasma concentrations of bupropion are
dose-proportional following single doses of 50 to 250 mg.
Following
oral administration of 200 mg 14C-bupropion HCL aqueous solution in
man, 81% and 10% of the radioactive dose were recovered over 48 hours in urine
and feces, respectively. the
fraction of the oral dose of bupropion excreted unchanged in urine was only
0.5%. It is extensively
metabolized by the liver. Plasma
and urinary metabolites are formed via reduction of the carbonyl group and/or
hydroxylation of the tert-butyl group of bupropion. Four basic
5
Back a Page
Next Page
Back to Wellbutrin SR. NDA Index Page
Back to Main
Index