DIVISION OF
NEUROPHARMACOLOGICAL DRUG PRODUCTS
Review of Chemistry,
Manufacturing, and Controls
(STAMPED: MAR 10 1995)
|
NDA#: 20-358 |
|
|||
|
CHEMISTRY REVIEW#: 2 |
DATE REVIEWED: 07-MAR-95 |
|||
|
SUBMISSION TYPE |
DOCUMENT DATE |
CDER
DATE |
ASSIGNED DATE |
|
|
ORIGINAL |
13-APR-93 |
13-APR-93 |
21-APR-93 |
|
|
RESUBMITTED |
28-FEB-94 |
28-FEB-94 |
|
|
|
AMENDMENT |
02-FEB-95 |
10-FEB-05 |
13-FEB-95 |
|
|
NAME & ADDRESS OF APPLICANT |
BURROUGHS WELLCOME COMPANY |
|||
|
|
3030 Cornwallis Road |
|||
|
|
Research Triangle Park, NC 27709 |
|||
|
|
|
|||
|
DRUG PRODUCT NAME |
|
|||
|
Proprietary: |
WELLBUTRIN® SR |
|||
|
Nonproprietary/Established/USAN: |
Bupropion hydrochloride |
|||
|
Code
Name/#: |
|
|||
|
Chem.
Type/Ther. Class |
|
|||
|
|
|
|||
|
ANDA Suitability Petition / DESI / Patent Status: |
N/A |
|||
|
PHARMACOLOGICAL CATEGORY/INDICATION: |
ANTIDEPRESSANT/DEPRESSION |
|||
|
|
|
|||
|
DOSAGE FORM: |
TABLETS |
|||
|
STRENGTHS: |
50, 100 and 150mg |
|||
|
ROUTE OF ADMINISTRATION: |
Oral |
|||
|
DISPENSED: |
XX Rx
___OTC |
|||
|
|
||||
|
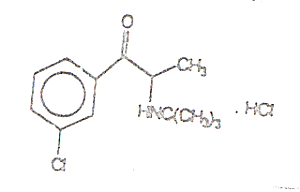
CHEMICAL NAME, STRUCTURAL FORMULA, MOLECULAR
FORMULA, MOLECULAR WEIGHT: |
||||
|
2-tert-butylamino-3’-chloropropiophenone
hydrochloride (±) |
|
|||
|
CAS #: 31677-93-7; 34911-55-2 (bupropion base) |
||||
|
Molecular Formula: C13H18CINO.
HCl |
||||
|
Moleculary Weight: 276.21 |
||||
|
|
WELLBUTRIN (Bupropion hydrochloride) |
|||
|
CONCLUSIONS & RECOMMENDATIONS: |
||||
|
Based on review of the Chemistry and Manufacturing
Controls Section of this submission, recommend that submission be considered UNAPPROVABLE
until validation of the regulatory test methods and satisfactory response to
chemist’s deficiency letter regarding expiry date are completed or
implemented. |
||||
cc:
Org. NDA 20-358
HFD-120/Division File
HFD-120/CPBarisek/3/8/95
HFD_120/David
HFD-120/SBlum
HFD-102/Ckumkumian [#1 only] (signature
dated 9/19/94)
Charles
B. Parisek, Ph.D., Review Chemist
filename: NO20358.002
R/D Init by: SBlum
Back a Page
Next Section Review of Environmental Assessment
Back to Wellbutrin SR® NDA Index Page
Back to Main
Index