DIVISION OF NEUROPHARMACOLOGICAL DRUG PRODUCTS
Review of Chemistry,
Manufacturing, and Controls
|
NDA#: 20-358 |
|
|||
|
CHEMISTRY REVIEW#: 4 |
DATE REVIEWED: 28-AUG-96 |
|||
|
SUBMISSION TYPE |
DOCUMENT DATE |
CDER
DATE |
ASSIGNED DATE |
|
|
AMENDMENT |
14-MAY-96 |
15-MAY-96 |
20-MAY-96 |
|
|
|
|
|
|
|
|
NAME & ADDRESS OF APPLICANT |
GLAXO WELLCOME INC |
|||
|
|
Five Moore Drive |
|||
|
|
Research Triangle Park, NC 27709 |
|||
|
|
|
|||
|
DRUG PRODUCT NAME |
|
|||
|
Proprietary: |
WELLBUTRIN SR |
|||
|
Nonproprietary/Established/USAN: |
Bupropion hydrochloride |
|||
|
Code
Name/#: |
|
|||
|
Chem.
Type/Ther. Class |
|
|||
|
PHARMACOLOGICAL CATEGORY/INDICATION: |
ANTIDEPRESSANT/DEPRESSION |
|||
|
DOSAGE FORM: |
TABLETS |
|||
|
STRENGTHS: |
50, 100 and 150mg |
|||
|
ROUTE OF ADMINISTRATION: |
Oral |
|||
|
DISPENSED: |
XX Rx
___OTC |
|||
|
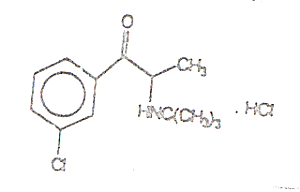
CHEMICAL NAME, STRUCTURAL FORMULA, MOLECULAR
FORMULA, MOLECULAR WEIGHT: |
||||
|
(±)-1-(3-chlorophenyl)-2-[1,1-dimethylethyl)amino]-1-propanone
hydrochloride (USAN) |
||||
|
CAS #: 31677-93-7; 34911-55-2 (bupropion base) |
||||
|
C13H18CINO. HCl Mol Wt:
276.21 |
||||
|
|
||||
|
|
||||
|
CONCLUSIONS & RECOMMENDATIONS: |
||||
|
based on review of the Chemistry and Manufacturing
Controls Section, we recommend that the the label storage statement revisions
and the addition of a cap liner to the container system be APPROVED; we do
not recommend approval of a 24 month |
|
|||
|
expiration date at this time or revision of the original dissolution specifications. |
||||
|
Methods Validation is still in progress. |
||||
cc:
Org.
NDA 20-358
HFD-120/Division
File
HFD-120/CBParisek/8/26/96(signature8/28/96)
HFD-120/David
HFD-120/SBlum
R/D
Init by: SBLUM
(Signature 8/29/96)
Charles B. Parisek, Ph.D., Review Chemist
Filename: NO20358a.004
Back a Page
Next Section - Non-Approval Letter
Back to Wellbutrin SR® NDA Index Page
Back to Main
Index